|
|
|
A cataract is a clouding of the eyes' natural lens. As we age, some clouding of the lens may occur. The first sign of a cataract is usually blurry vision. Although glasses and other visual aids may at first help a person with cataracts, surgery may become inevitable. Cataract surgery is very successful in restoring vision, and it is the most frequently performed surgery in the United States.
Approximately 500,000 babies are born each year in the United States to teenage mothers.
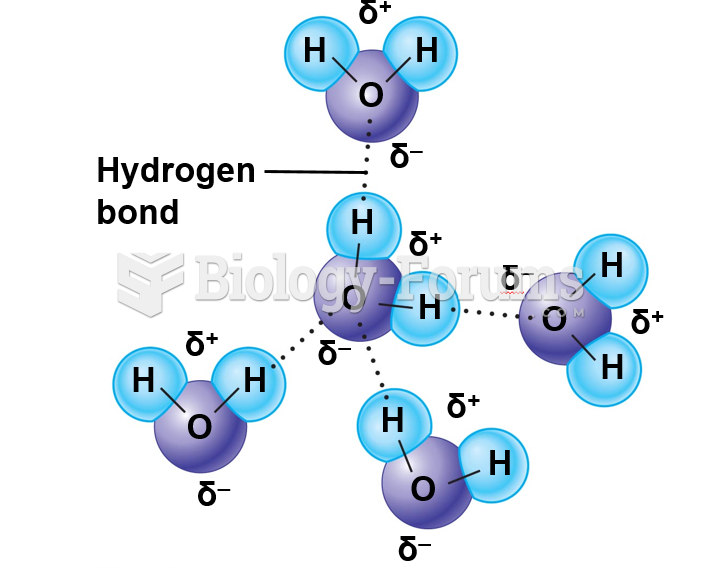
The ratio of hydrogen atoms to oxygen in water (H2O) is 2:1.
It is widely believed that giving a daily oral dose of aspirin to heart attack patients improves their chances of survival because the aspirin blocks the formation of new blood clots.
For pediatric patients, intravenous fluids are the most commonly cited products involved in medication errors that are reported to the USP.