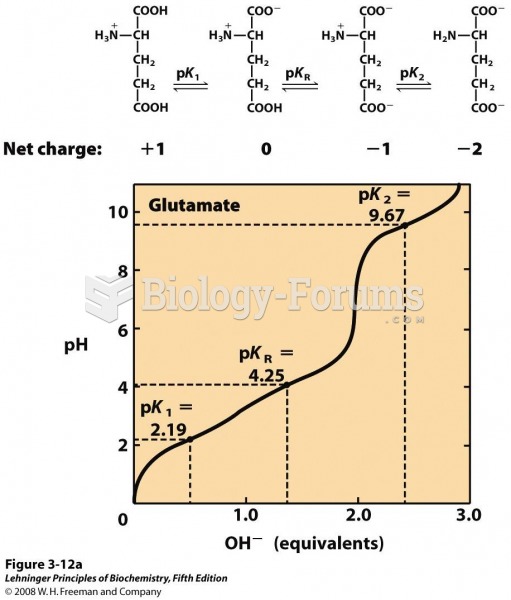
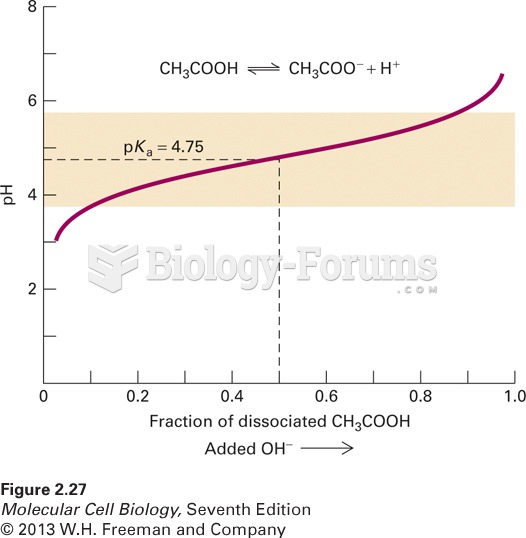
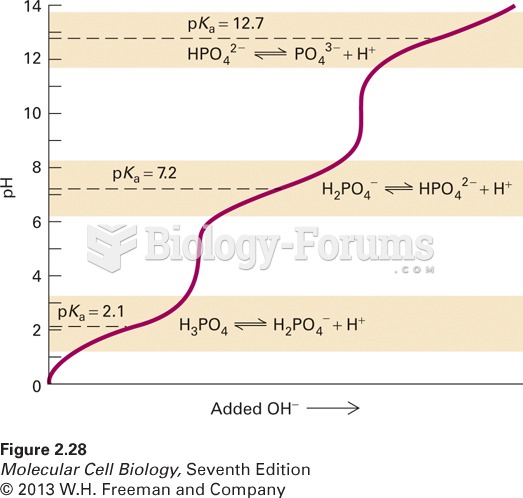
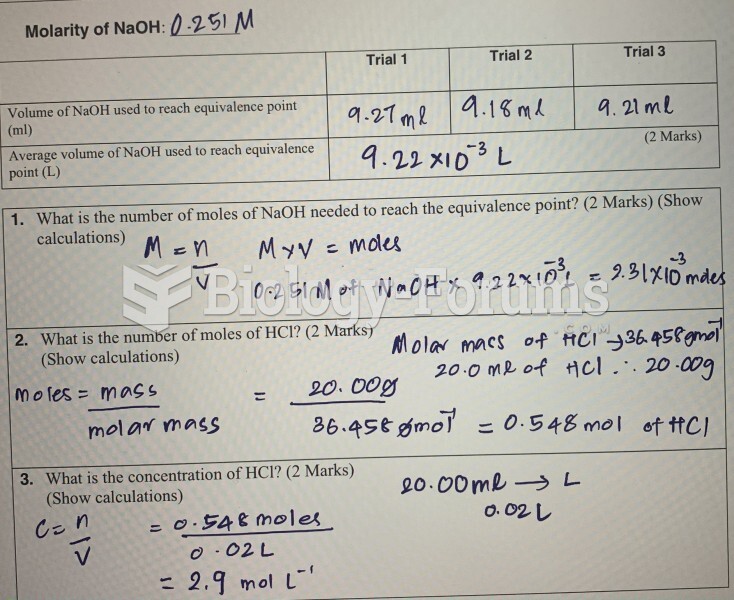
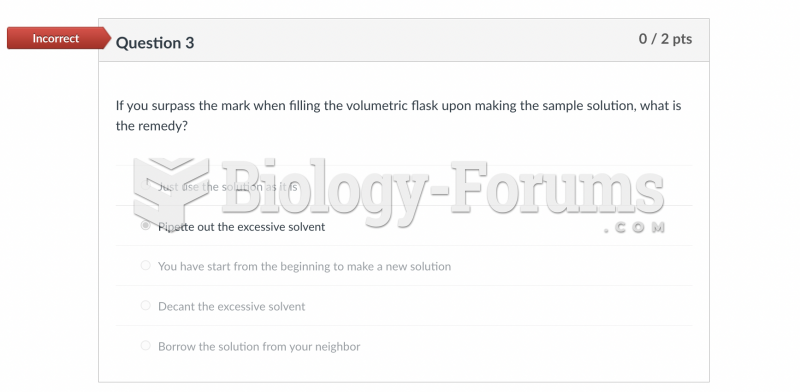
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
If all the neurons in the human body were lined up, they would stretch more than 600 miles.
Did you know?
Women are 50% to 75% more likely than men to experience an adverse drug reaction.
Did you know?
People with high total cholesterol have about two times the risk for heart disease as people with ideal levels.
Did you know?
About 100 new prescription or over-the-counter drugs come into the U.S. market every year.
Did you know?
The effects of organophosphate poisoning are referred to by using the abbreviations “SLUD” or “SLUDGE,” It stands for: salivation, lacrimation, urination, defecation, GI upset, and emesis.