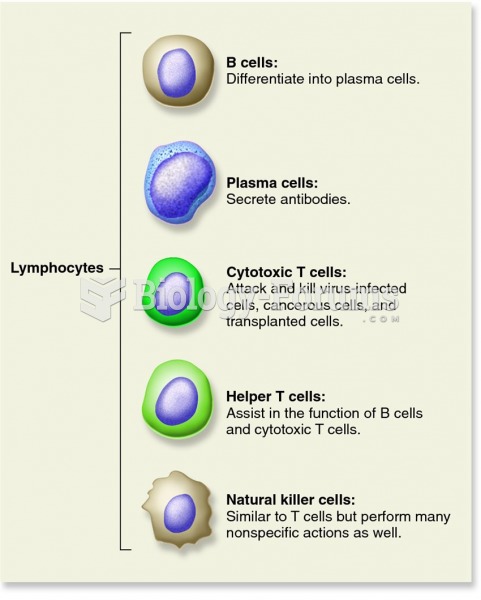
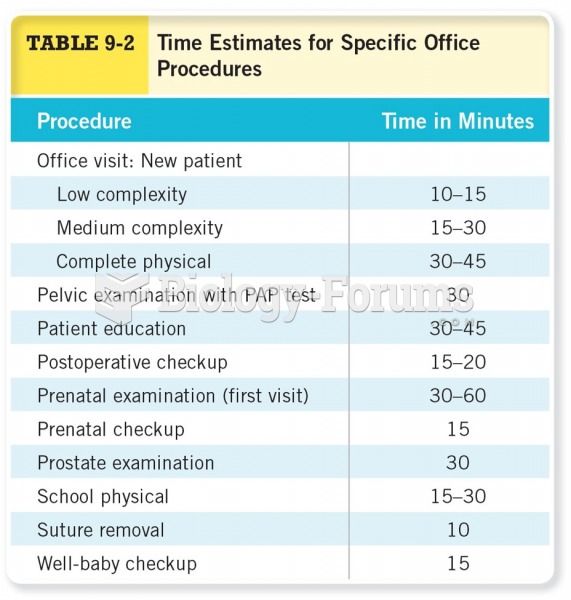
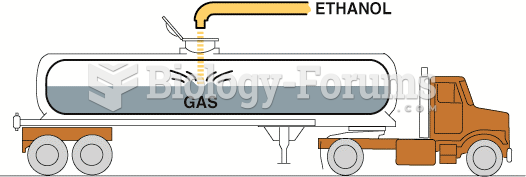
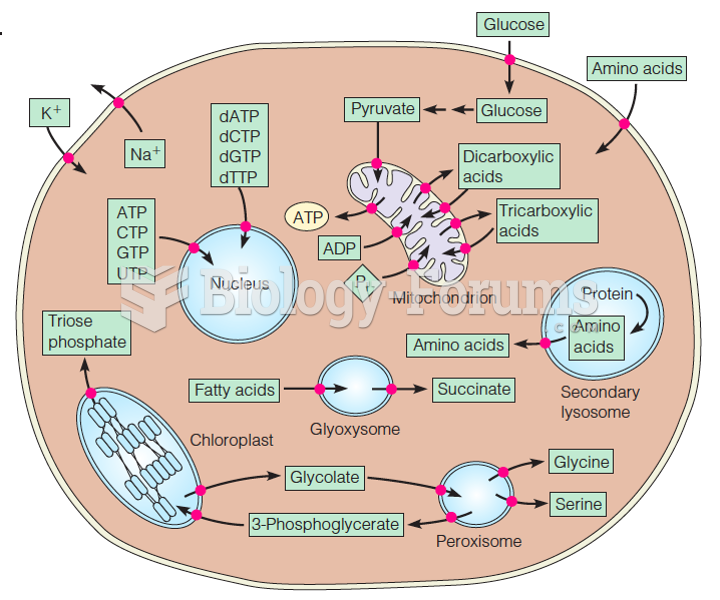
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
The heart is located in the center of the chest, with part of it tipped slightly so that it taps against the left side of the chest.
Did you know?
HIV testing reach is still limited. An estimated 40% of people with HIV (more than 14 million) remain undiagnosed and do not know their infection status.
Did you know?
There are 20 feet of blood vessels in each square inch of human skin.
Did you know?
Human kidneys will clean about 1 million gallons of blood in an average lifetime.
Did you know?
The senior population grows every year. Seniors older than 65 years of age now comprise more than 13% of the total population. However, women outlive men. In the 85-and-over age group, there are only 45 men to every 100 women.