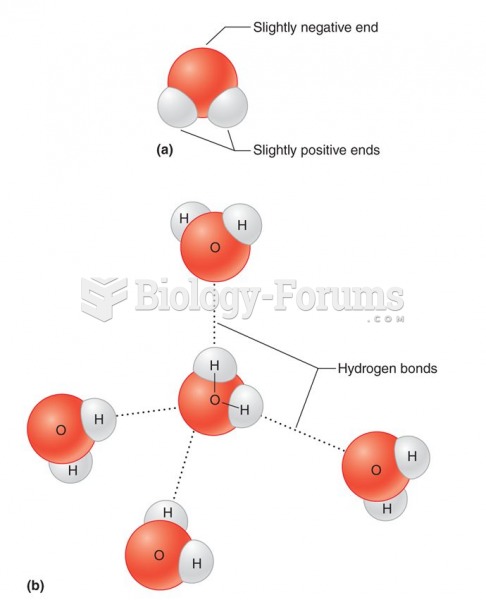
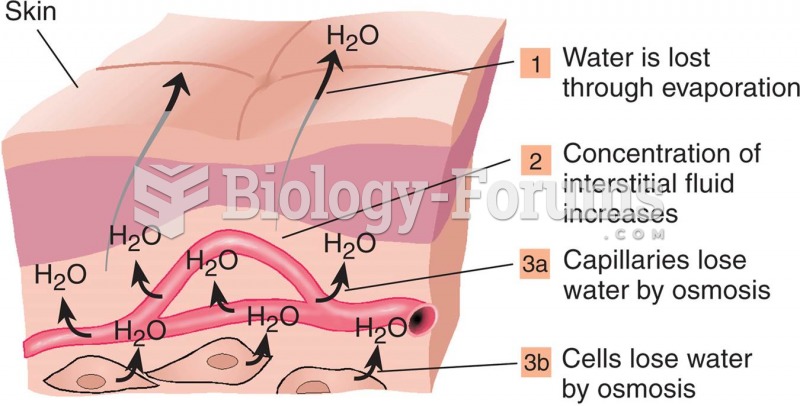
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
By definition, when a medication is administered intravenously, its bioavailability is 100%.
Did you know?
Human kidneys will clean about 1 million gallons of blood in an average lifetime.
Did you know?
People about to have surgery must tell their health care providers about all supplements they take.
Did you know?
The first war in which wide-scale use of anesthetics occurred was the Civil War, and 80% of all wounds were in the extremities.
Did you know?
Interferon was scarce and expensive until 1980, when the interferon gene was inserted into bacteria using recombinant DNA technology, allowing for mass cultivation and purification from bacterial cultures.