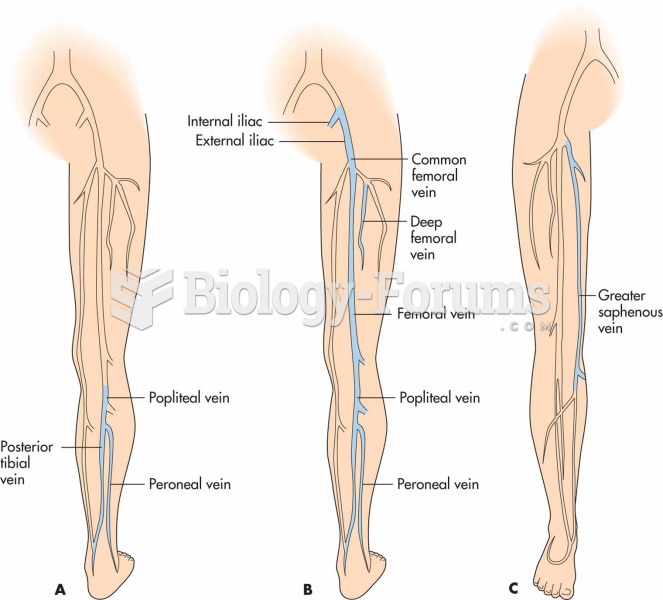
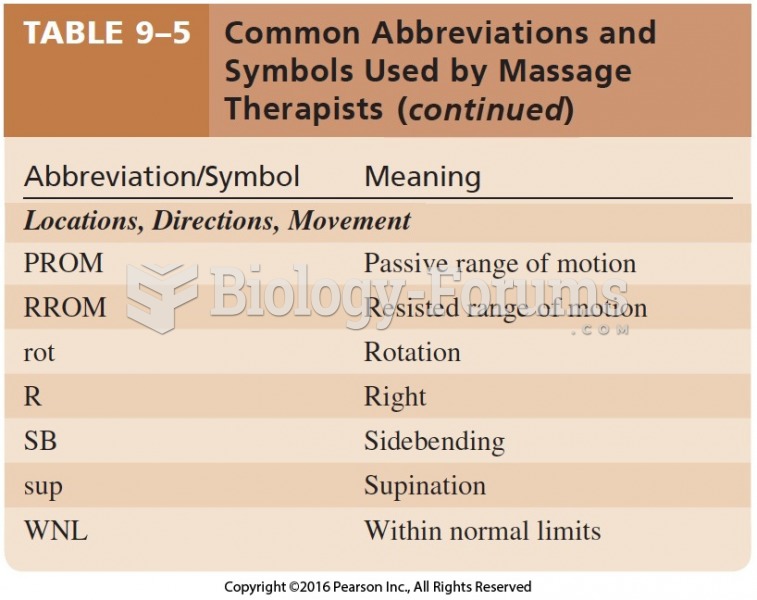
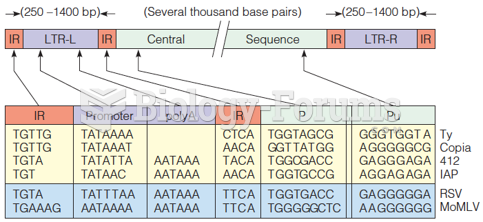
|
|
|
During the twentieth century, a variant of the metric system was used in Russia and France in which the base unit of mass was the tonne. Instead of kilograms, this system used millitonnes (mt).
In 1864, the first barbiturate (barbituric acid) was synthesized.
Egg cells are about the size of a grain of sand. They are formed inside of a female's ovaries before she is even born.
Although the Roman numeral for the number 4 has always been taught to have been "IV," according to historians, the ancient Romans probably used "IIII" most of the time. This is partially backed up by the fact that early grandfather clocks displayed IIII for the number 4 instead of IV. Early clockmakers apparently thought that the IIII balanced out the VIII (used for the number 8) on the clock face and that it just looked better.
Drying your hands with a paper towel will reduce the bacterial count on your hands by 45–60%.