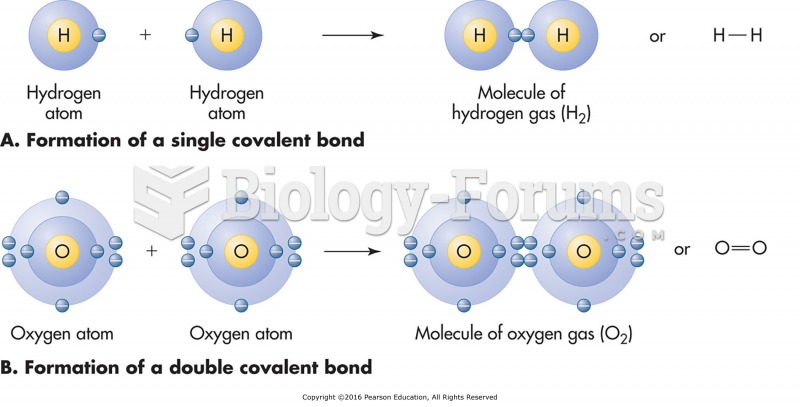
Following is a chair conformation of cyclohexane with the carbon atoms numbered 1 through 6.

(a) Draw hydrogen atoms that are above the plane of the ring on carbons 1 and 2 and below the plane of the ring on carbon
4.
(b) Which of these hydrogens are equatorial? Which are axial?
(c) Draw the alternative chair conformation. Which hydrogens are equatorial? Which are axial? Which are above the plane
of the ring? Which are below it?
Question 2Write molecular formulas for each bicycloalkane, given its number of carbon atoms.
(a) Hydrindane (9 carbons)
(b) Decalin (10 carbons)
(c) Norbornane (7 carbons)
Question 3Write the molecular formula, IUPAC name, and common name for each cycloalkane.


 Question 4
Question 4Combine the proper prefix, infix, and suffix and write the IUPAC name for each compound.
Question 5Draw line-angle formulas for the three constitutional isomers with the molecular formula C5H12.
Question 6Do the line-angle formulas in each pair represent the same compound or constitutional isomers.

 Question 7
Question 7Describe the bonding in PCl
5 without using
d orbitals. As a hint, the geometry of PCl
5 is as shown:
Question 8/nA4eGN/5LNzV39aeDDlOTl1H6zWnVPX6ndmWJWp0YsOr/cnaNfdv3kw3Txdut3AxYCrs/wwiDMUpP2dJwTWnUPK7eyPnCfXzkuZzj/F6K+jbyp7qrDs7yGp7wa0FZoBEWK5AAABw0lEQVQZHtUn/ih35dkbDy8X2A5wqRH6RMkK1Kg4k/lUDXbmsOL3Z7oG4dIPhhOuM1Jry80X2mg52K8yHGYp/18ahxlaLRoCyxiBwTEpp3oP24ne7l6v1z8euKY3MbFm8t30R/cGR/YG6Uweuxp1PC9+ExO9Nck3EfJu+8HaKLsnUi5l6uaxpremHnHrl7U3FG8Zt+jIqvfbcoBVQgwwOwy9dh6EGXF87nBbTD0mOp1/P5tum2nLfvopz8Rk+87xuN8Uuu3EHS53/k/0jwOi8dQ3NNP/n2OHg7oNsKl9Ys2aSqeV9ocwm1L3Ndt7faoNwEvbXaF6S1nBaXVrCCxfBHIUq93DvnxpYL5qPqCtsr0cbMs58wXyvKfblgDmHeKWQUPgZCFw5AbAk1WSlu9SQ6BPW0csJS21ai7x+rQZgCXewK16DYGGQEOgIdAQGIVAmwEYhUrzawg0BBoCDYGGwBJHoCkAS7yBW/UaAg2BhkBDoCEwCoGmAIxCpfk1BBoCDYGGQENgiSPQFIAl3sCteg2BhkBDoCHQEBiFQFMARqHS/BoCDYGGQEOgIbDEEWgKwBJv4Fa9hkBDoCHQEGgIjELg/wP4RDh/hWZHXQAAAABJRU5ErkJggg== />
Based on the bond angles
Question 9Why are the following molecular formulas impossible?
(a) CH
5 (b) C2H7