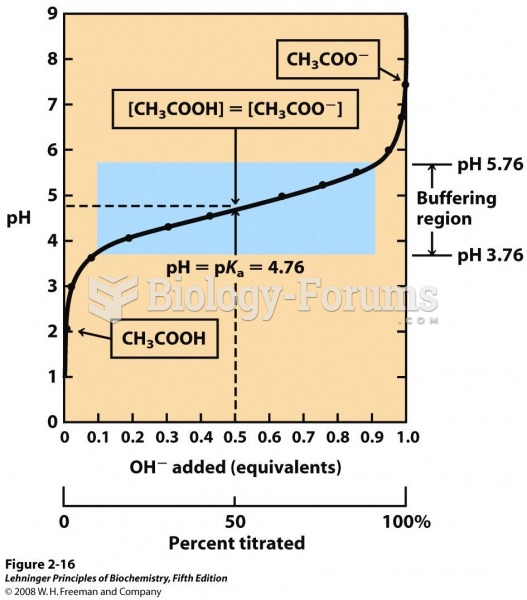
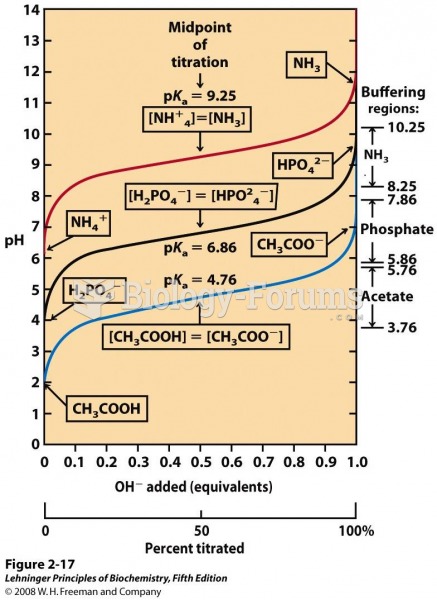
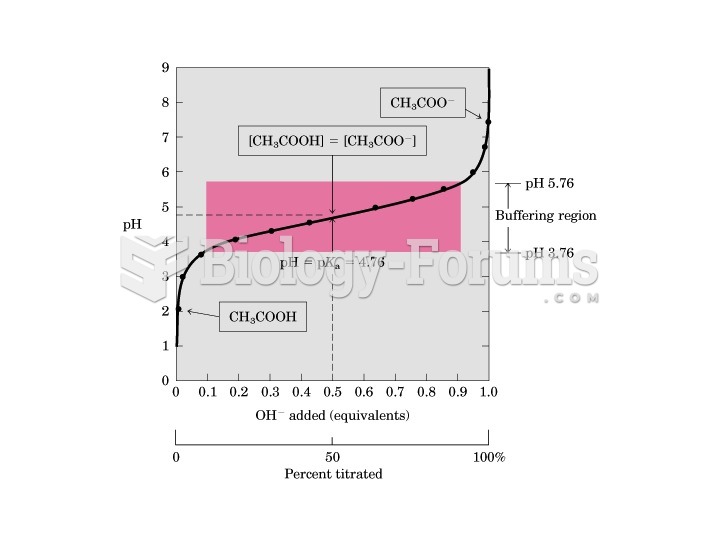
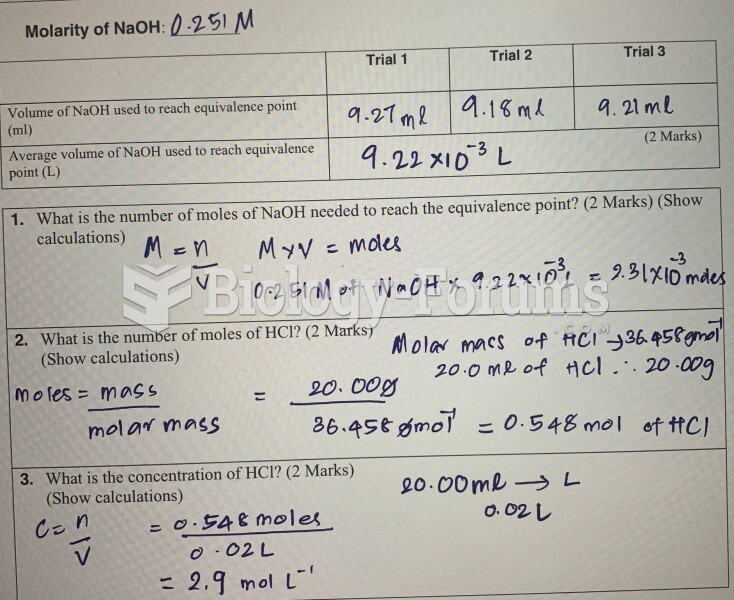
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Approximately 500,000 babies are born each year in the United States to teenage mothers.
Did you know?
There are more sensory neurons in the tongue than in any other part of the body.
Did you know?
In 1885, the Lloyd Manufacturing Company of Albany, New York, promoted and sold "Cocaine Toothache Drops" at 15 cents per bottle! In 1914, the Harrison Narcotic Act brought the sale and distribution of this drug under federal control.
Did you know?
Intradermal injections are somewhat difficult to correctly administer because the skin layers are so thin that it is easy to accidentally punch through to the deeper subcutaneous layer.
Did you know?
The ratio of hydrogen atoms to oxygen in water (H2O) is 2:1.