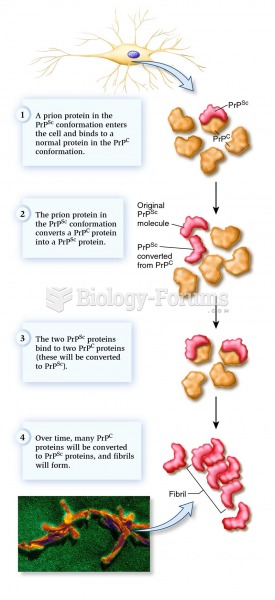
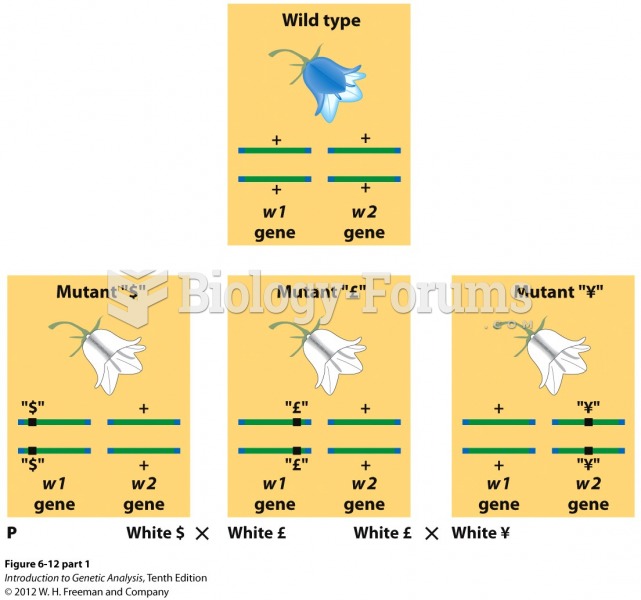
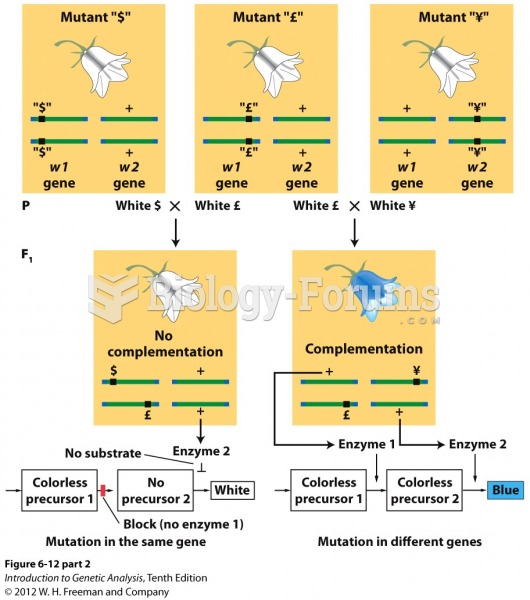
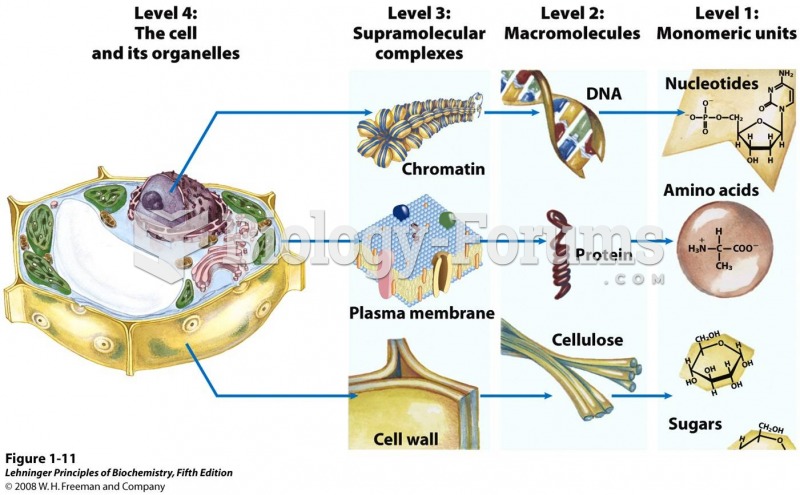
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Fungal nail infections account for up to 30% of all skin infections. They affect 5% of the general population—mostly people over the age of 70.
Did you know?
About one in five American adults and teenagers have had a genital herpes infection—and most of them don't know it. People with genital herpes have at least twice the risk of becoming infected with HIV if exposed to it than those people who do not have genital herpes.
Did you know?
There are more bacteria in your mouth than there are people in the world.
Did you know?
In 1844, Charles Goodyear obtained the first patent for a rubber condom.
Did you know?
The first oncogene was discovered in 1970 and was termed SRC (pronounced "SARK").