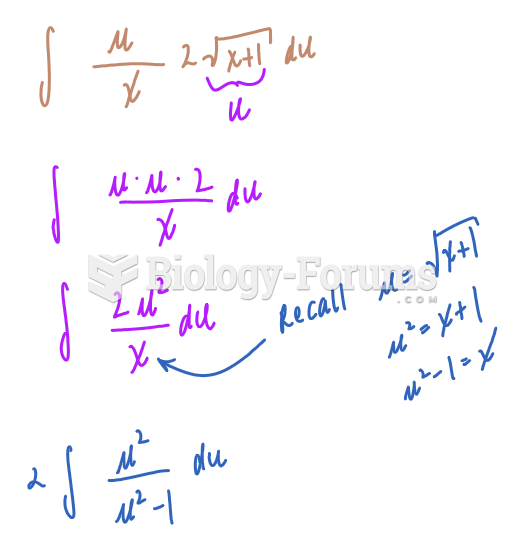|
|
|
Children with strabismus (crossed eyes) can be treated. They are not able to outgrow this condition on their own, but with help, it can be more easily corrected at a younger age. It is important for infants to have eye examinations as early as possible in their development and then another at age 2 years.
Ether was used widely for surgeries but became less popular because of its flammability and its tendency to cause vomiting. In England, it was quickly replaced by chloroform, but this agent caused many deaths and lost popularity.
Human kidneys will clean about 1 million gallons of blood in an average lifetime.
People about to have surgery must tell their health care providers about all supplements they take.
A seasonal flu vaccine is the best way to reduce the chances you will get seasonal influenza and spread it to others.