|
|
|
Multiple experimental evidences have confirmed that at the molecular level, cancer is caused by lesions in cellular DNA.
As the western states of America were settled, pioneers often had to drink rancid water from ponds and other sources. This often resulted in chronic diarrhea, causing many cases of dehydration and death that could have been avoided if clean water had been available.
According to the CDC, approximately 31.7% of the U.S. population has high low-density lipoprotein (LDL) or "bad cholesterol" levels.
Bacteria have flourished on the earth for over three billion years. They were the first life forms on the planet.
People with alcoholism are at a much greater risk of malnutrition than are other people and usually exhibit low levels of most vitamins (especially folic acid). This is because alcohol often takes the place of 50% of their daily intake of calories, with little nutritional value contained in it.
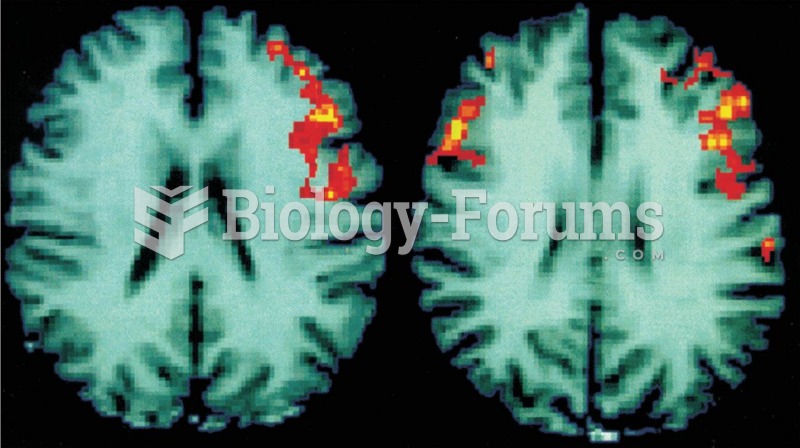 Functional MRI Scans These scans of human brains show localized average increases in neural activity
Functional MRI Scans These scans of human brains show localized average increases in neural activity
 Piaget was one of the first to go beyond observable behavior in an attempt to determine how infants ...
Piaget was one of the first to go beyond observable behavior in an attempt to determine how infants ...