This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
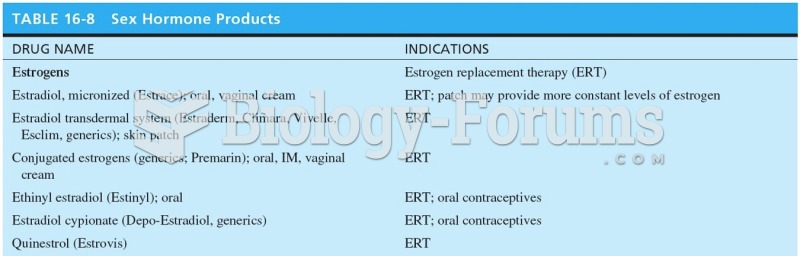
It is important to read food labels and choose foods with low cholesterol and saturated trans fat. You should limit saturated fat to no higher than 6% of daily calories.
Did you know?
Approximately 500,000 babies are born each year in the United States to teenage mothers.
Did you know?
Patients should never assume they are being given the appropriate drugs. They should make sure they know which drugs are being prescribed, and always double-check that the drugs received match the prescription.
Did you know?
The first monoclonal antibodies were made exclusively from mouse cells. Some are now fully human, which means they are likely to be safer and may be more effective than older monoclonal antibodies.
Did you know?
The immune system needs 9.5 hours of sleep in total darkness to recharge completely.