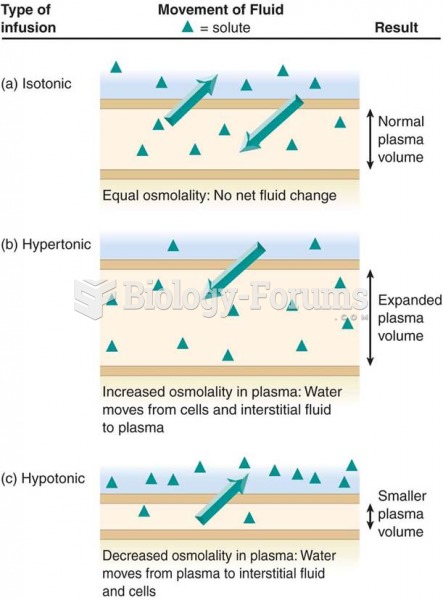
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Medication errors are more common among seriously ill patients than with those with minor conditions.
Did you know?
Congestive heart failure is a serious disorder that carries a reduced life expectancy. Heart failure is usually a chronic illness, and it may worsen with infection or other physical stressors.
Did you know?
On average, the stomach produces 2 L of hydrochloric acid per day.
Did you know?
This year, an estimated 1.4 million Americans will have a new or recurrent heart attack.
Did you know?
More than 4.4billion prescriptions were dispensed within the United States in 2016.