|
|
|
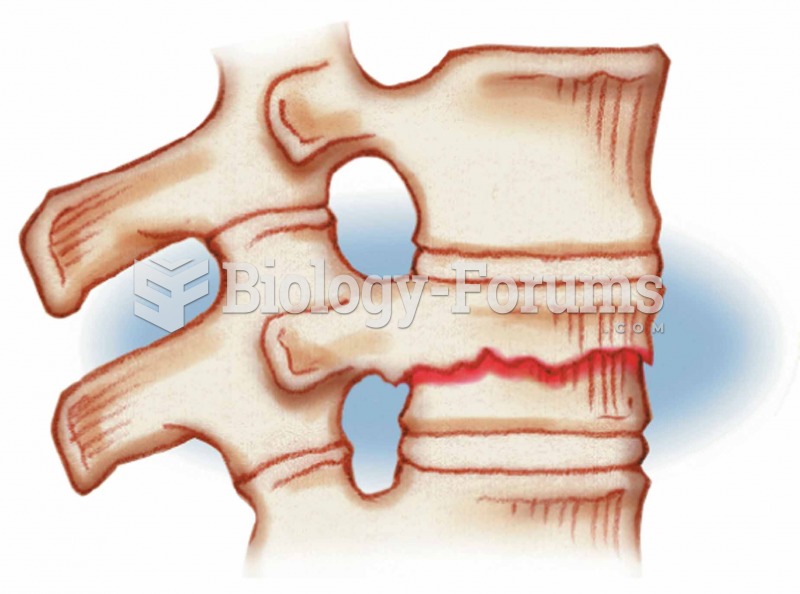
There are immediate benefits of chiropractic adjustments that are visible via magnetic resonance imaging (MRI). It shows that spinal manipulation therapy is effective in decreasing pain and increasing the gaps between the vertebrae, reducing pressure that leads to pain.
The horizontal fraction bar was introduced by the Arabs.
If you use artificial sweeteners, such as cyclamates, your eyes may be more sensitive to light. Other factors that will make your eyes more sensitive to light include use of antibiotics, oral contraceptives, hypertension medications, diuretics, and antidiabetic medications.
Illicit drug use costs the United States approximately $181 billion every year.
Blood in the urine can be a sign of a kidney stone, glomerulonephritis, or other kidney problems.