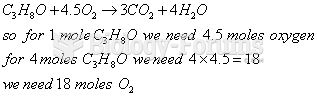|
|
|
Fewer than 10% of babies are born on their exact due dates, 50% are born within 1 week of the due date, and 90% are born within 2 weeks of the date.
People with high total cholesterol have about two times the risk for heart disease as people with ideal levels.
On average, someone in the United States has a stroke about every 40 seconds. This is about 795,000 people per year.
The first oncogene was discovered in 1970 and was termed SRC (pronounced "SARK").
As many as 28% of hospitalized patients requiring mechanical ventilators to help them breathe (for more than 48 hours) will develop ventilator-associated pneumonia. Current therapy involves intravenous antibiotics, but new antibiotics that can be inhaled (and more directly treat the infection) are being developed.