This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Though “Krazy Glue” or “Super Glue” has the ability to seal small wounds, it is not recommended for this purpose since it contains many substances that should not enter the body through the skin, and may be harmful.
Did you know?
Between 1999 and 2012, American adults with high total cholesterol decreased from 18.3% to 12.9%
Did you know?
Less than one of every three adults with high LDL cholesterol has the condition under control. Only 48.1% with the condition are being treated for it.
Did you know?
Green tea is able to stop the scent of garlic or onion from causing bad breath.
Did you know?
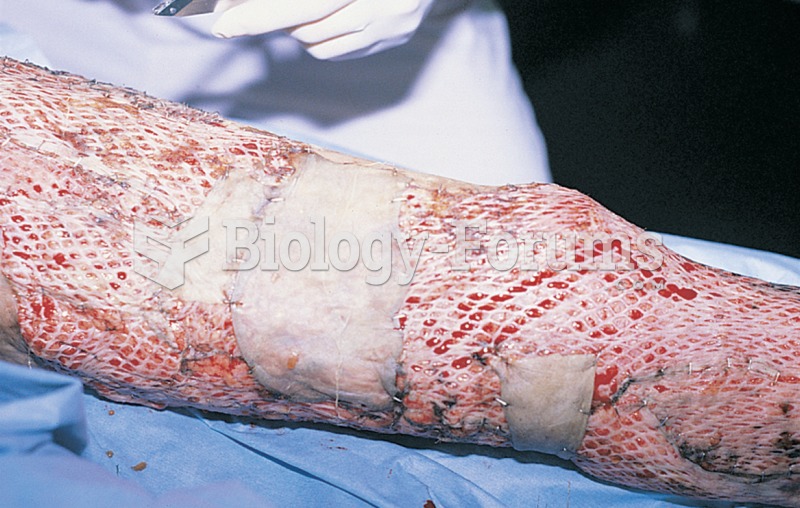
There are 20 feet of blood vessels in each square inch of human skin.