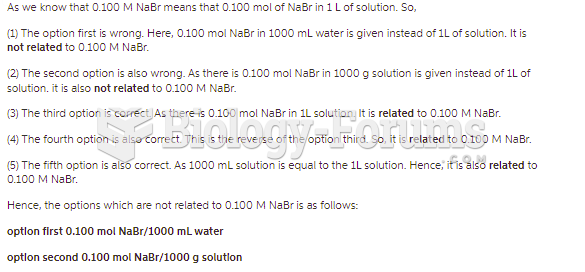
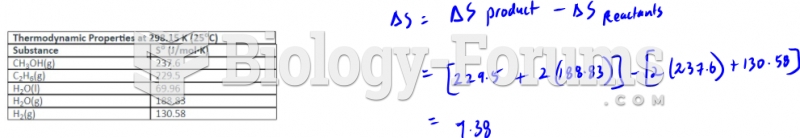This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
The familiar sounds of your heart are made by the heart's valves as they open and close.
Did you know?
Anti-aging claims should not ever be believed. There is no supplement, medication, or any other substance that has been proven to slow or stop the aging process.
Did you know?
Green tea is able to stop the scent of garlic or onion from causing bad breath.
Did you know?
The modern decimal position system was the invention of the Hindus (around 800 AD), involving the placing of numerals to indicate their value (units, tens, hundreds, and so on).
Did you know?
Each year in the United States, there are approximately six million pregnancies. This means that at any one time, about 4% of women in the United States are pregnant.