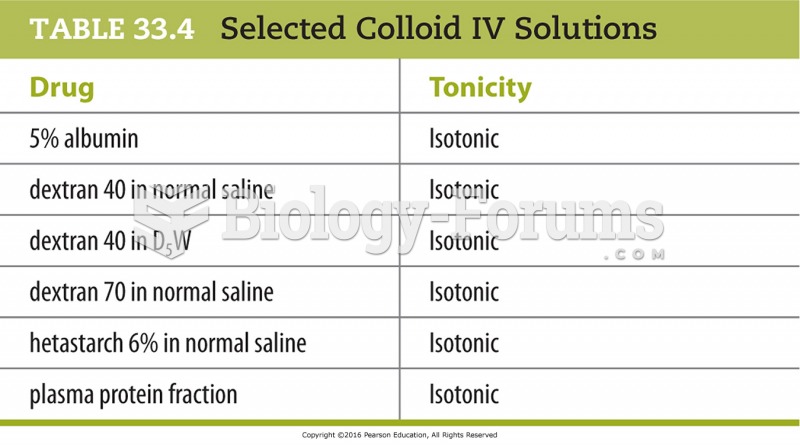
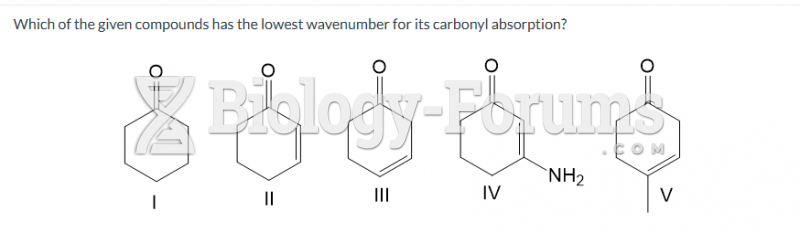
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Approximately 15–25% of recognized pregnancies end in miscarriage. However, many miscarriages often occur before a woman even knows she is pregnant.
Did you know?
Excessive alcohol use costs the country approximately $235 billion every year.
Did you know?
Eating carrots will improve your eyesight. Carrots are high in vitamin A (retinol), which is essential for good vision. It can also be found in milk, cheese, egg yolks, and liver.
Did you know?
Blood is approximately twice as thick as water because of the cells and other components found in it.
Did you know?
The familiar sounds of your heart are made by the heart's valves as they open and close.