This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Green tea is able to stop the scent of garlic or onion from causing bad breath.
Did you know?
The word drug comes from the Dutch word droog (meaning "dry"). For centuries, most drugs came from dried plants, hence the name.
Did you know?
The B-complex vitamins and vitamin C are not stored in the body and must be replaced each day.
Did you know?
Serum cholesterol testing in adults is recommended every 1 to 5 years. People with diabetes and a family history of high cholesterol should be tested even more frequently.
Did you know?
Cytomegalovirus affects nearly the same amount of newborns every year as Down syndrome.
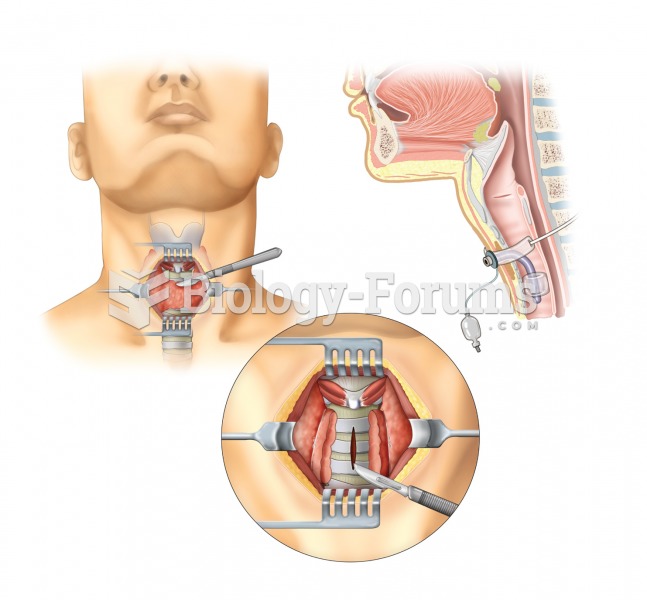 Tracheostomy. A tracheotomy, or incision into the trachea, is performed to create an opening into th
Tracheostomy. A tracheotomy, or incision into the trachea, is performed to create an opening into th
 The Kingsley plantation, on Fort George Island in Jacksonville, Florida. Zephaniah Kingsley, the own
The Kingsley plantation, on Fort George Island in Jacksonville, Florida. Zephaniah Kingsley, the own