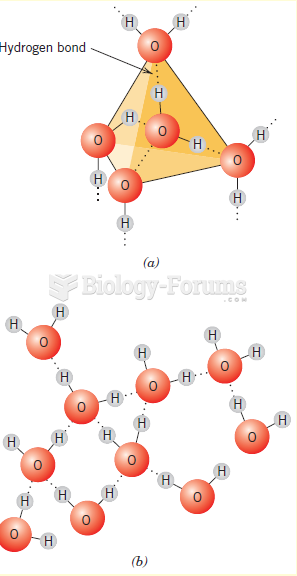
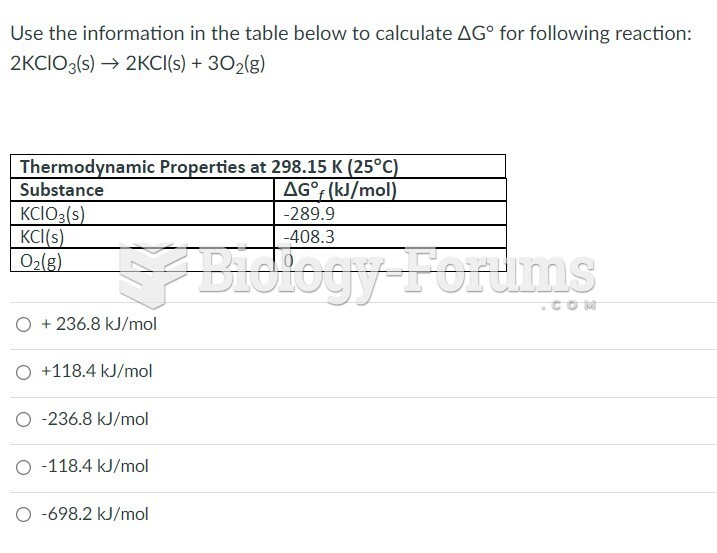
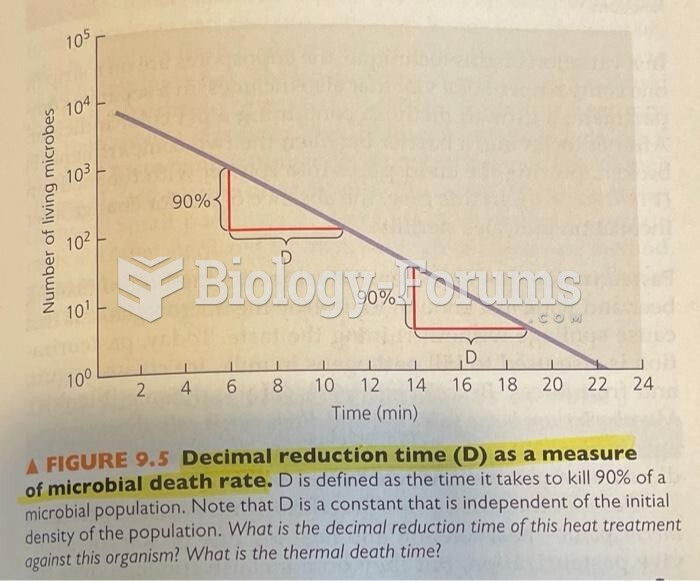
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
About 100 new prescription or over-the-counter drugs come into the U.S. market every year.
Did you know?
Multiple experimental evidences have confirmed that at the molecular level, cancer is caused by lesions in cellular DNA.
Did you know?
HIV testing reach is still limited. An estimated 40% of people with HIV (more than 14 million) remain undiagnosed and do not know their infection status.
Did you know?
More than 50% of American adults have oral herpes, which is commonly known as "cold sores" or "fever blisters." The herpes virus can be active on the skin surface without showing any signs or causing any symptoms.
Did you know?
Approximately 500,000 babies are born each year in the United States to teenage mothers.