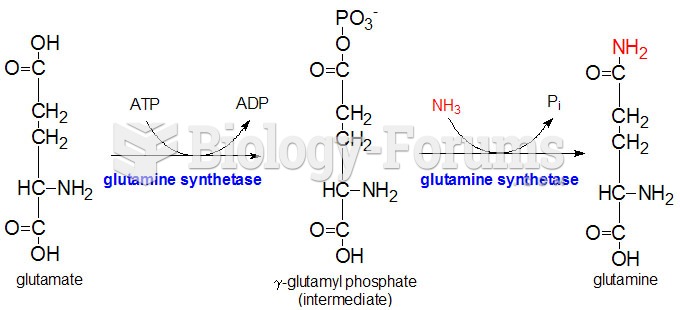
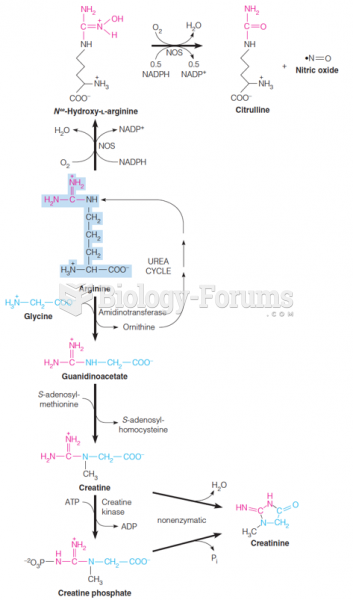
|
|
|
Chronic marijuana use can damage the white blood cells and reduce the immune system's ability to respond to disease by as much as 40%. Without a strong immune system, the body is vulnerable to all kinds of degenerative and infectious diseases.
More than 2,500 barbiturates have been synthesized. At the height of their popularity, about 50 were marketed for human use.
More than 34,000 trademarked medication names and more than 10,000 generic medication names are in use in the United States.
About 100 new prescription or over-the-counter drugs come into the U.S. market every year.
Although puberty usually occurs in the early teenage years, the world's youngest parents were two Chinese children who had their first baby when they were 8 and 9 years of age.