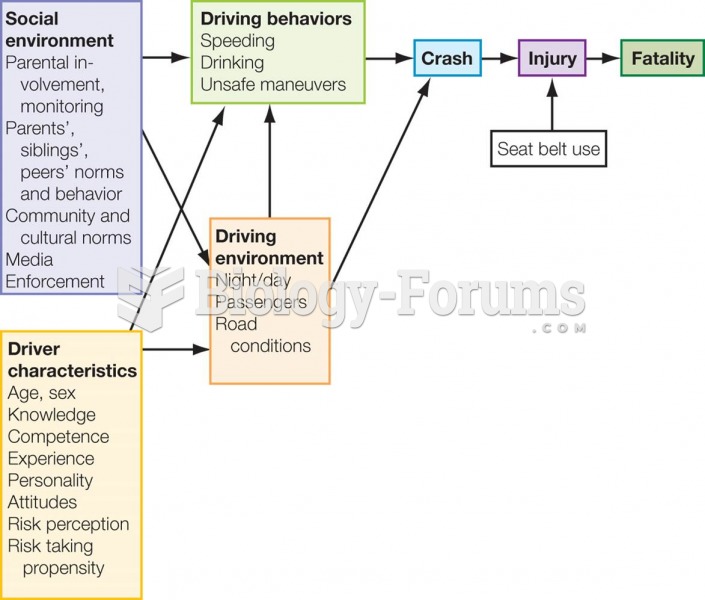
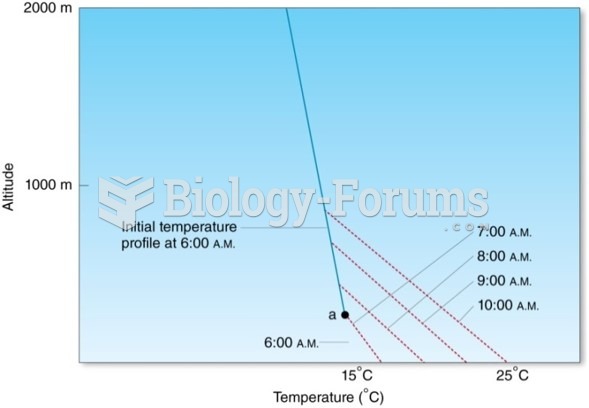
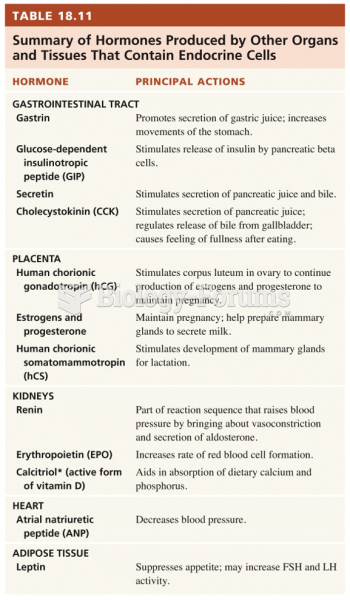
Answer to Question 1
Iron occurs in two forms in foods: as heme iron, bound into the iron-carrying proteins such as hemoglobin in meats, poultry, and fish; and as nonheme iron, in both plant and animal foods. Heme iron is much more reliably absorbed than is nonheme iron. Absorption of nonheme iron is affected by many factors, including the amount of vitamin C consumed with meals. Vitamin C promotes iron absorption and can triple the amount of nonheme iron absorbed from foods eaten at the same meal.
Contamination ironthat is, iron obtained from cookware or soilcan also increase iron intake significantly. Consumers who cook their foods in iron cookware thereby contribute to their iron intake. For example, the iron content of a half-cup of spaghetti sauce simmered in a glass dish is 3 milligrams, but it is 87 milligrams when the sauce is cooked in an iron skillet. Similarly, dried apricots and raisins contain more iron than the fresh fruit because they are dried in iron pans. Admittedly, this form of iron is not as well absorbed as the iron from meat, but every little bit helps.
Some foods contain binders that combine chemically with iron and other minerals, such as calcium and zinc, thus preventing absorption of those substances and carrying them out of the body with other wastes. For example, phytates render the iron, calcium, zinc, and magnesium in certain foods less available than they might otherwise be; oxalate binds iron and calcium. Phytate is found in oatmeal and other whole grains. Other binders are the tannins, which occur in black teas, coffee, cola drinks, chocolate, and red wines. Fiber also can reduce iron absorption because it speeds up the transit of materials through the intestines.
Answer to Question 2
D