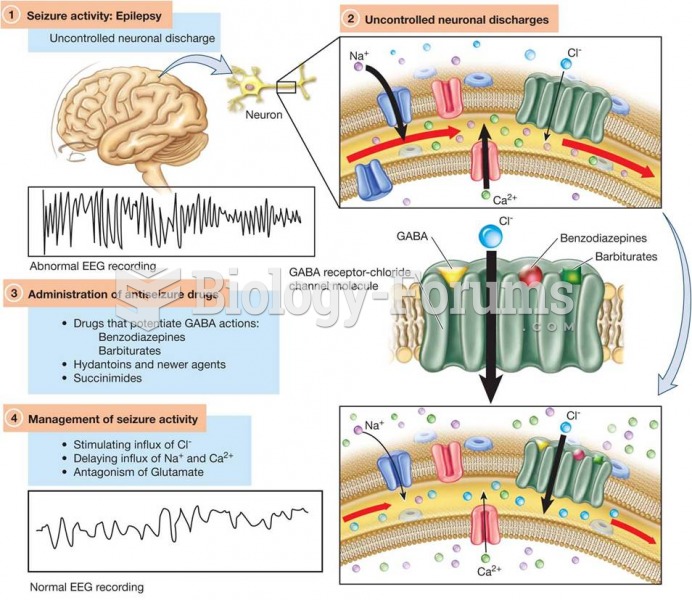
|
|
|
Eat fiber! A diet high in fiber can help lower cholesterol levels by as much as 10%.
To maintain good kidney function, you should drink at least 3 quarts of water daily. Water dilutes urine and helps prevent concentrations of salts and minerals that can lead to kidney stone formation. Chronic dehydration is a major contributor to the development of kidney stones.
Autoimmune diseases occur when the immune system destroys its own healthy tissues. When this occurs, white blood cells cannot distinguish between pathogens and normal cells.
Side effects from substance abuse include nausea, dehydration, reduced productivitiy, and dependence. Though these effects usually worsen over time, the constant need for the substance often overcomes rational thinking.
There used to be a metric calendar, as well as metric clocks. The metric calendar, or "French Republican Calendar" divided the year into 12 months, but each month was divided into three 10-day weeks. Each day had 10 decimal hours. Each hour had 100 decimal minutes. Due to lack of popularity, the metric clocks and calendars were ended in 1795, three years after they had been first marketed.
 The skeleton of a dwarf elephant from the island of Crete. Dwarf elephants were present on some Medi
The skeleton of a dwarf elephant from the island of Crete. Dwarf elephants were present on some Medi
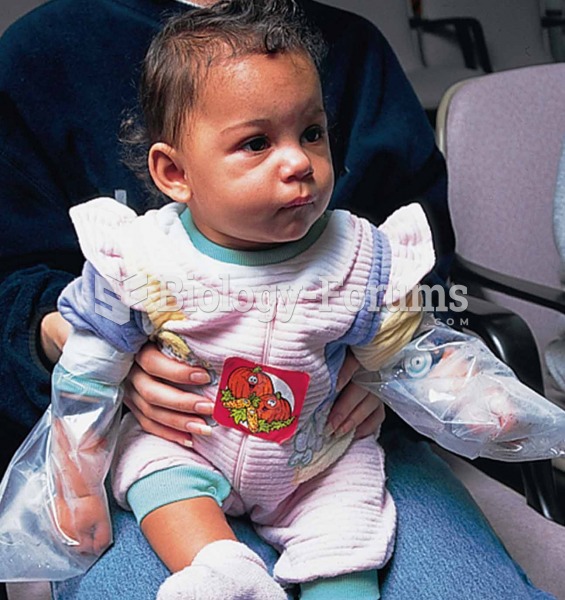 Evaluation of a child for cystic fibrosis with a sweat chloride test. Sweat is being collected under
Evaluation of a child for cystic fibrosis with a sweat chloride test. Sweat is being collected under