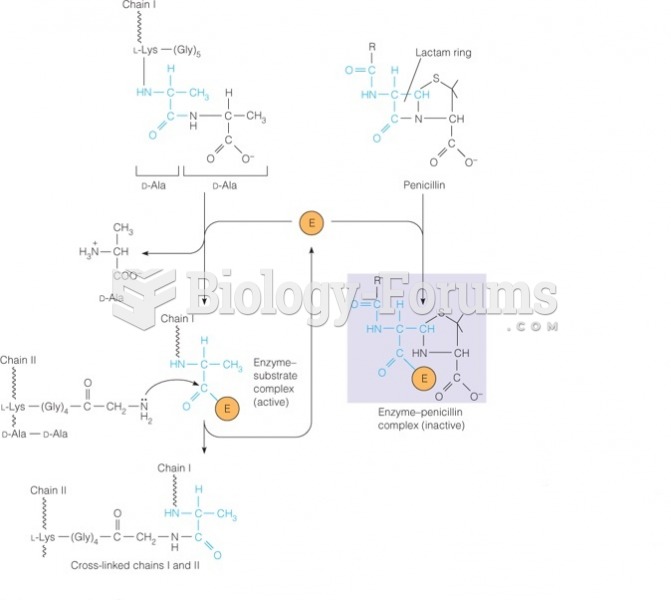
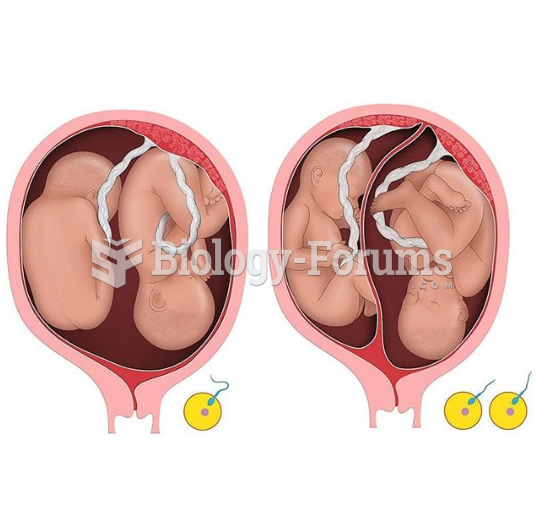
|
|
|
The highest suicide rate in the United States is among people ages 65 years and older. Almost 15% of people in this age group commit suicide every year.
Astigmatism is the most common vision problem. It may accompany nearsightedness or farsightedness. It is usually caused by an irregularly shaped cornea, but sometimes it is the result of an irregularly shaped lens. Either type can be corrected by eyeglasses, contact lenses, or refractive surgery.
Common abbreviations that cause medication errors include U (unit), mg (milligram), QD (every day), SC (subcutaneous), TIW (three times per week), D/C (discharge or discontinue), HS (at bedtime or "hours of sleep"), cc (cubic centimeters), and AU (each ear).
Medication errors are more common among seriously ill patients than with those with minor conditions.
If you use artificial sweeteners, such as cyclamates, your eyes may be more sensitive to light. Other factors that will make your eyes more sensitive to light include use of antibiotics, oral contraceptives, hypertension medications, diuretics, and antidiabetic medications.
 Forest fragments left by clear-cutting forest from the surrounding landscape have very different phy
Forest fragments left by clear-cutting forest from the surrounding landscape have very different phy
 The “Big Four” world leaders meet at the Hotel Crillon in Paris, 1919. From left to right (front row
The “Big Four” world leaders meet at the Hotel Crillon in Paris, 1919. From left to right (front row