Answer to Question 1
Correct Answer: 4
Rationale 1: The maturity of the liver of newborns was not a reason the recombinant form of hepatitis B vaccine was developed.
Rationale 2: A recombinant form was not developed due to increased allergic response.
Rationale 3: Virus inactivation was not the driving concern for development of the recombinant form.
Rationale 4: A recombinant form was developed due to fear of HIV transmission.
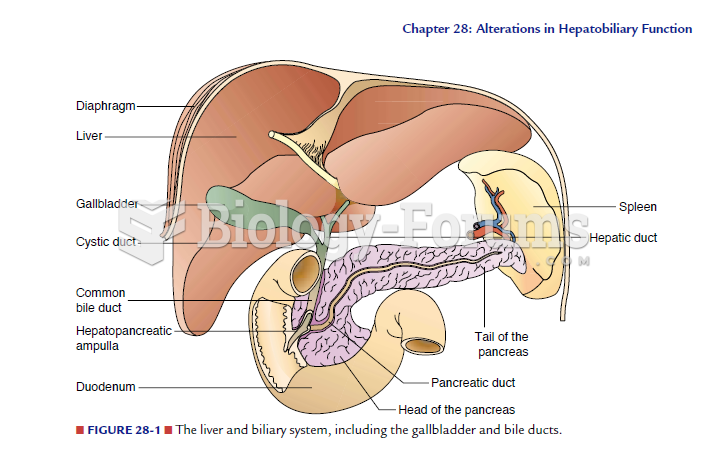
Global Rationale: Perhaps the best example of the use of this form of biotechnology is for the development of the hepatitis B immunization. It was originally obtained from the serum of infected individuals but carried the risk of transmitting HIV when it was administered as a vaccine. Through advances in recombinant deoxyribonucleic acid (DNA) technology, yeast cells have been reengineered to produce proteins very similar to those found on the coating of the hepatitis B virus, allowing for the development of safe immune protection from this virus. Liver maturity, allergic responses, and virus inactivation were not consideration for this development.
Answer to Question 2
Correct Answer: 4
Rationale 1: These agents are examples of toxin vaccines.
Rationale 2: Measles and mumps are not whole-agent vaccines.
Rationale 3: These are examples of subunit vaccines.
Rationale 4: Whole-agent vaccines include influenza and hepatitis A.
Global Rationale: Whole-agent vaccines consist of microbes killed by heat or chemicals. Inactivated or killed vaccines are safer than live vaccines because the organism is unable to replicate, mutate, or cause disease. Subsequent booster doses are required to maintain immunity if inactivated or killed vaccines are used. Examples of inactivated or killed vaccines include the influenza and hepatitis A vaccines. Diphtheria and tetanus are toxin vaccines. Measles and mumps are not whole-agent vaccines. HPV and pertussis are subunit vaccines.