This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Cytomegalovirus affects nearly the same amount of newborns every year as Down syndrome.
Did you know?
Children with strabismus (crossed eyes) can be treated. They are not able to outgrow this condition on their own, but with help, it can be more easily corrected at a younger age. It is important for infants to have eye examinations as early as possible in their development and then another at age 2 years.
Did you know?
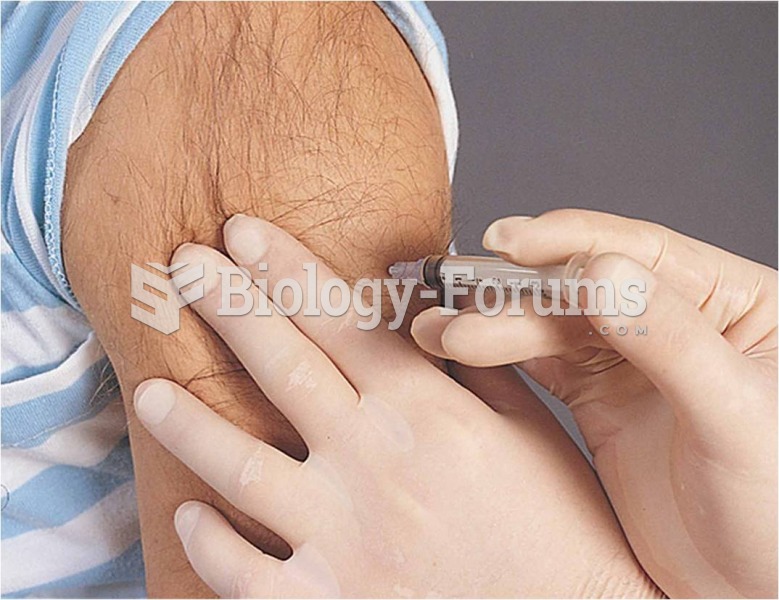
People about to have surgery must tell their health care providers about all supplements they take.
Did you know?
About 100 new prescription or over-the-counter drugs come into the U.S. market every year.
Did you know?
Recent studies have shown that the number of medication errors increases in relation to the number of orders that are verified per pharmacist, per work shift.