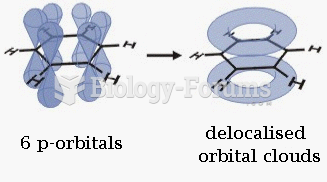
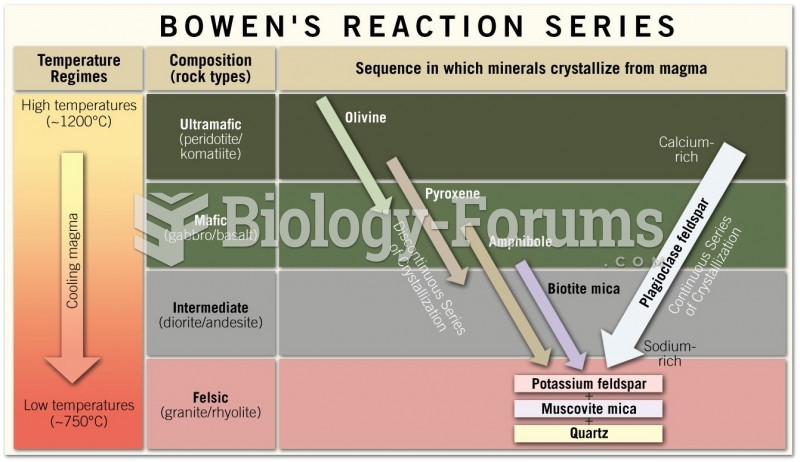
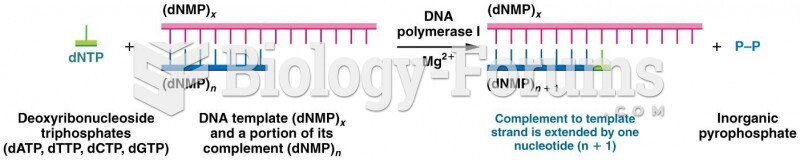
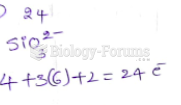|
|
|
The ratio of hydrogen atoms to oxygen in water (H2O) is 2:1.
More than 4.4billion prescriptions were dispensed within the United States in 2016.
Human kidneys will clean about 1 million gallons of blood in an average lifetime.
About 80% of major fungal systemic infections are due to Candida albicans. Another form, Candida peritonitis, occurs most often in postoperative patients. A rare disease, Candida meningitis, may follow leukemia, kidney transplant, other immunosuppressed factors, or when suffering from Candida septicemia.
As many as 28% of hospitalized patients requiring mechanical ventilators to help them breathe (for more than 48 hours) will develop ventilator-associated pneumonia. Current therapy involves intravenous antibiotics, but new antibiotics that can be inhaled (and more directly treat the infection) are being developed.