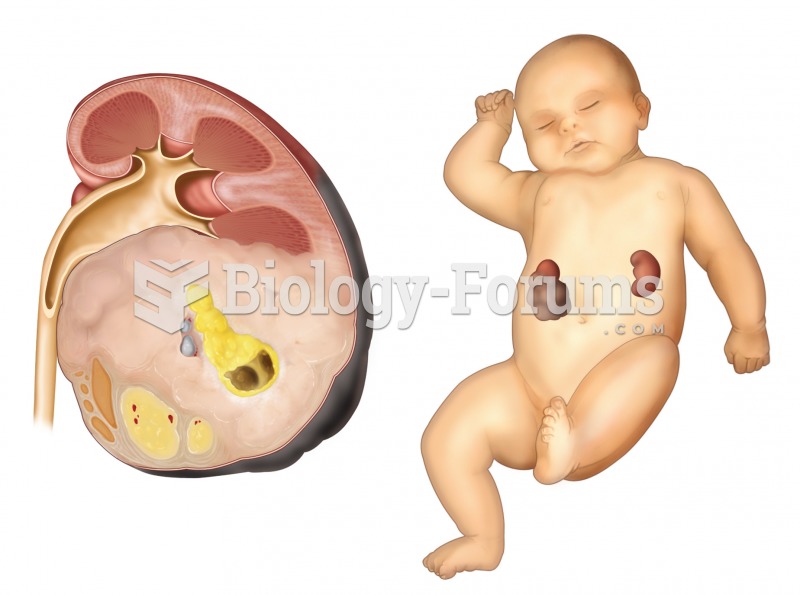
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Children with strabismus (crossed eyes) can be treated. They are not able to outgrow this condition on their own, but with help, it can be more easily corrected at a younger age. It is important for infants to have eye examinations as early as possible in their development and then another at age 2 years.
Did you know?
Approximately 70% of expectant mothers report experiencing some symptoms of morning sickness during the first trimester of pregnancy.
Did you know?
There are 60,000 miles of blood vessels in every adult human.
Did you know?
Medication errors are more common among seriously ill patients than with those with minor conditions.
Did you know?
For pediatric patients, intravenous fluids are the most commonly cited products involved in medication errors that are reported to the USP.