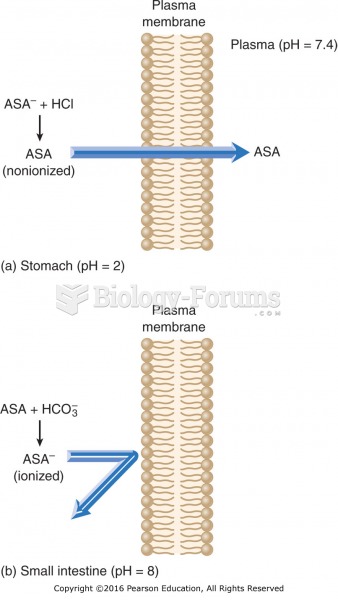
|
|
|
Studies show that systolic blood pressure can be significantly lowered by taking statins. In fact, the higher the patient's baseline blood pressure, the greater the effect of statins on his or her blood pressure.
Prostaglandins were first isolated from human semen in Sweden in the 1930s. They were so named because the researcher thought that they came from the prostate gland. In fact, prostaglandins exist and are synthesized in almost every cell of the body.
The modern decimal position system was the invention of the Hindus (around 800 AD), involving the placing of numerals to indicate their value (units, tens, hundreds, and so on).
In 2010, opiate painkllers, such as morphine, OxyContin®, and Vicodin®, were tied to almost 60% of drug overdose deaths.
The average office desk has 400 times more bacteria on it than a toilet.
 Several species of squirrels have melanistic phases. In large parts of United States and Canada, the
Several species of squirrels have melanistic phases. In large parts of United States and Canada, the
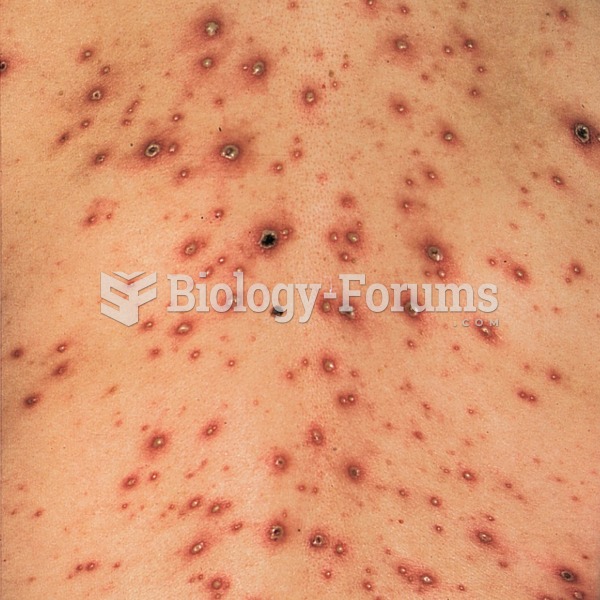 Varicella or chickenpox, a viral skin infection. In this photograph, the rash is beginning to form s
Varicella or chickenpox, a viral skin infection. In this photograph, the rash is beginning to form s