|
|
|
The first oral chemotherapy drug for colon cancer was approved by FDA in 2001.
Not getting enough sleep can greatly weaken the immune system. Lack of sleep makes you more likely to catch a cold, or more difficult to fight off an infection.
There are more bacteria in your mouth than there are people in the world.
According to the FDA, adverse drug events harmed or killed approximately 1,200,000 people in the United States in the year 2015.
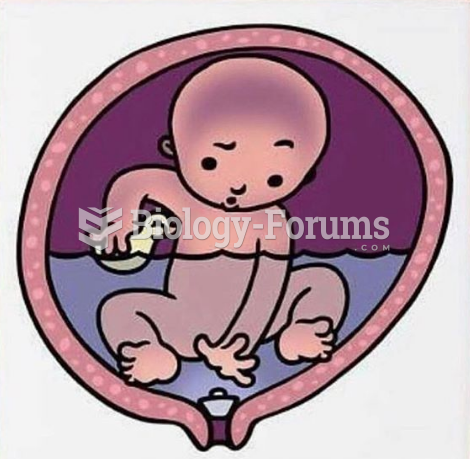
A serious new warning has been established for pregnant women against taking ACE inhibitors during pregnancy. In the study, the risk of major birth defects in children whose mothers took ACE inhibitors during the first trimester was nearly three times higher than in children whose mothers didn't take ACE inhibitors. Physicians can prescribe alternative medications for pregnant women who have symptoms of high blood pressure.