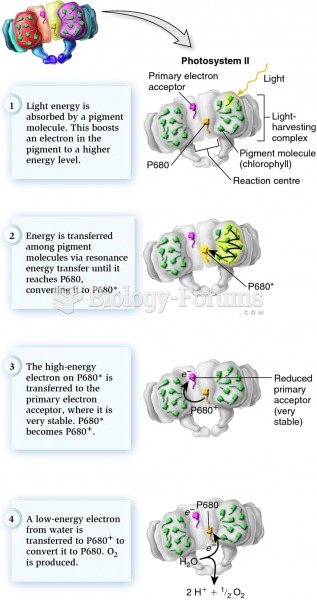
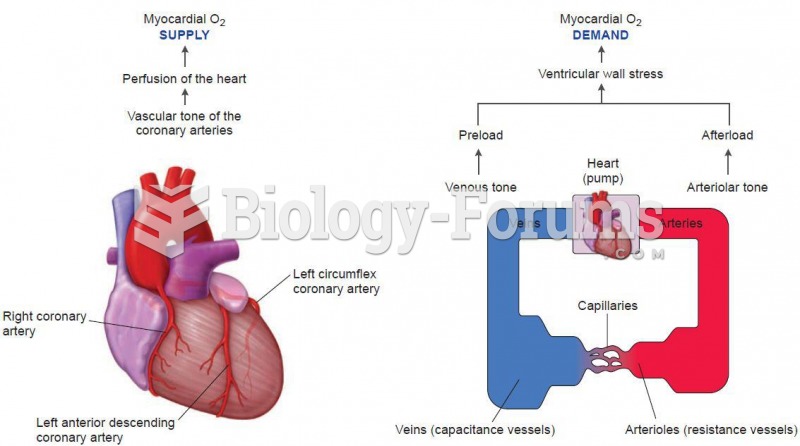
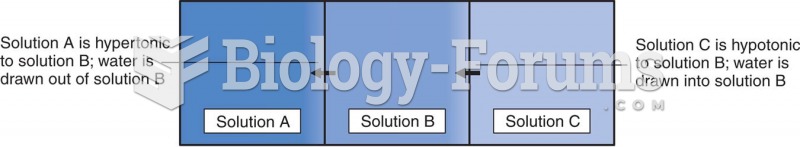
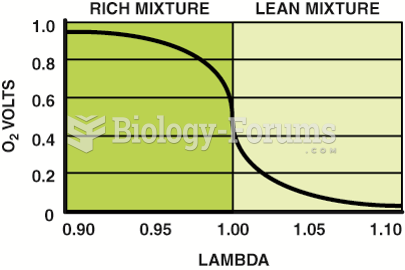
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Less than one of every three adults with high LDL cholesterol has the condition under control. Only 48.1% with the condition are being treated for it.
Did you know?
The word drug comes from the Dutch word droog (meaning "dry"). For centuries, most drugs came from dried plants, hence the name.
Did you know?
More than 20 million Americans cite use of marijuana within the past 30 days, according to the National Survey on Drug Use and Health (NSDUH). More than 8 million admit to using it almost every day.
Did you know?
Green tea is able to stop the scent of garlic or onion from causing bad breath.
Did you know?
More than 34,000 trademarked medication names and more than 10,000 generic medication names are in use in the United States.