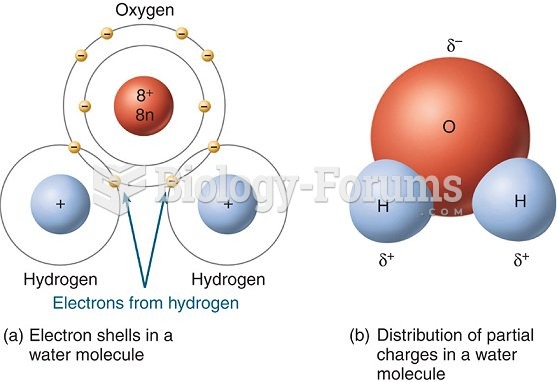
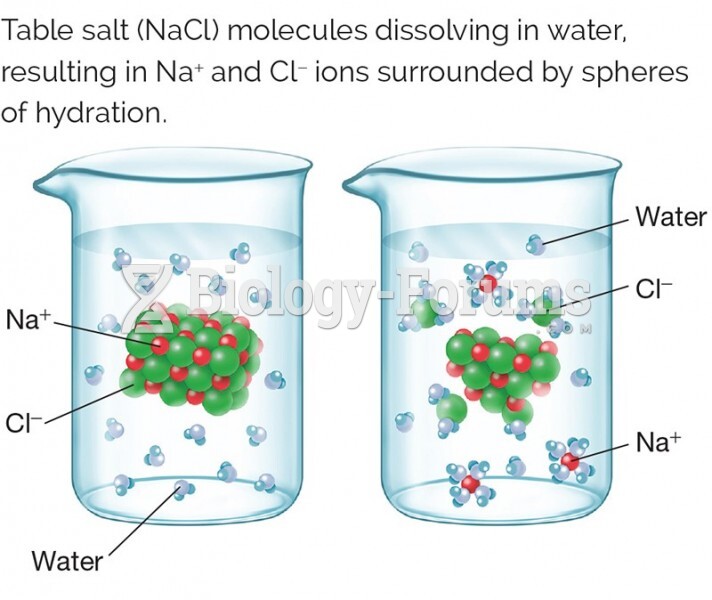
|
|
|
Computer programs are available that crosscheck a new drug's possible trade name with all other trade names currently available. These programs detect dangerous similarities between names and alert the manufacturer of the drug.
In the United States, an estimated 50 million unnecessary antibiotics are prescribed for viral respiratory infections.
As many as 20% of Americans have been infected by the fungus known as Histoplasmosis. While most people are asymptomatic or only have slight symptoms, infection can progress to a rapid and potentially fatal superinfection.
Although puberty usually occurs in the early teenage years, the world's youngest parents were two Chinese children who had their first baby when they were 8 and 9 years of age.
When intravenous medications are involved in adverse drug events, their harmful effects may occur more rapidly, and be more severe than errors with oral medications. This is due to the direct administration into the bloodstream.
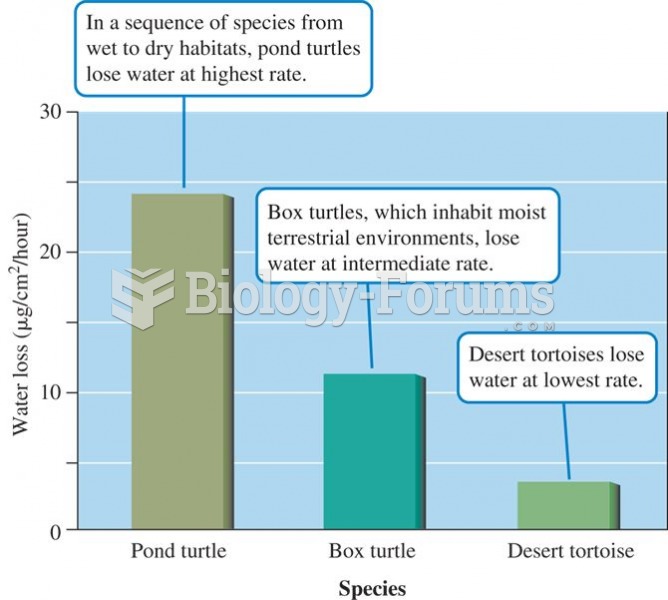 Rates of water loss by two turtles and a tortoise indicate an inverse relationship between the dryne
Rates of water loss by two turtles and a tortoise indicate an inverse relationship between the dryne
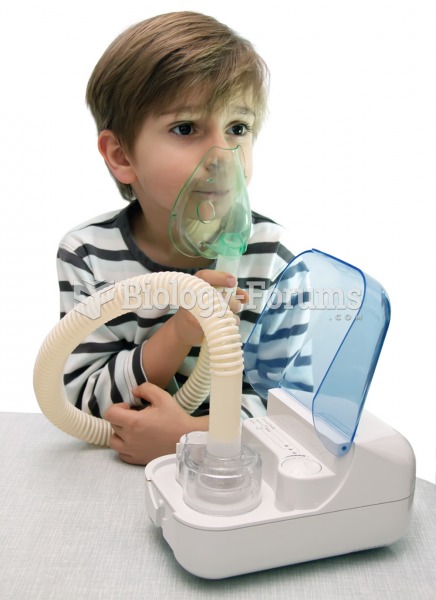 Nebulizer. The nebulizer converts a liquid medication to a mist that is easily inhaled. A face mask,
Nebulizer. The nebulizer converts a liquid medication to a mist that is easily inhaled. A face mask,