|
|
|
Did you know?
Elderly adults are at greatest risk of stroke and myocardial infarction and have the most to gain from prophylaxis. Patients ages 60 to 80 years with blood pressures above 160/90 mm Hg should benefit from antihypertensive treatment.
Did you know?
Today, nearly 8 out of 10 pregnant women living with HIV (about 1.1 million), receive antiretrovirals.
Did you know?
In ancient Rome, many of the richer people in the population had lead-induced gout. The reason for this is unclear. Lead poisoning has also been linked to madness.
Did you know?
Malaria was not eliminated in the United States until 1951. The term eliminated means that no new cases arise in a country for 3 years.
Did you know?
The familiar sounds of your heart are made by the heart's valves as they open and close.


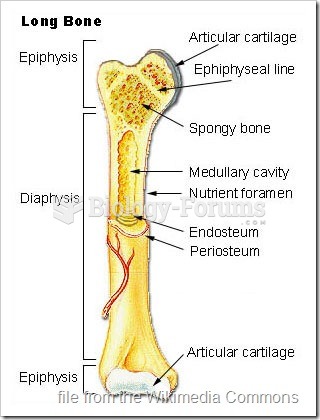

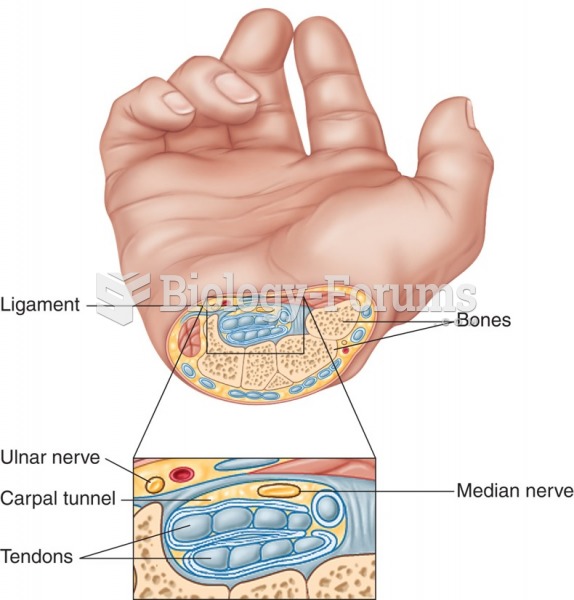
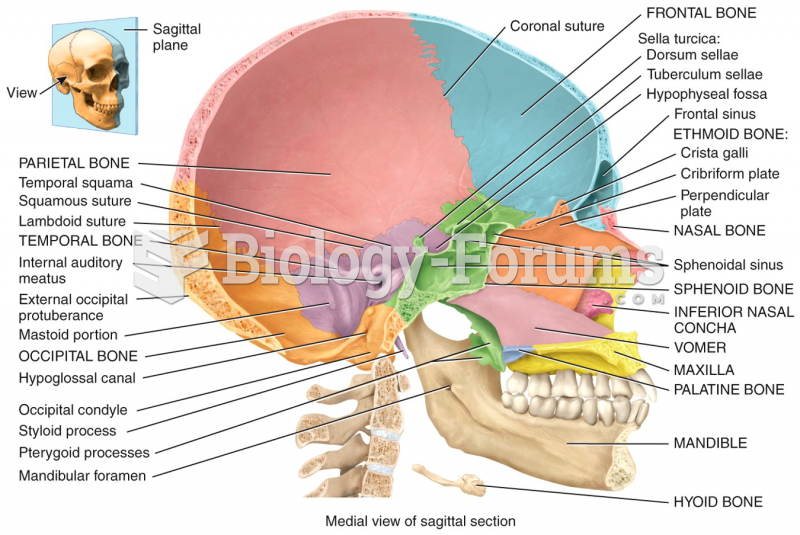
![Bolier - Best Friend (Never Let Me Down) [Official Music Video]](https://biology-forums.com/gallery/47/medium_6_01_10_23_10_03_00.jpg)