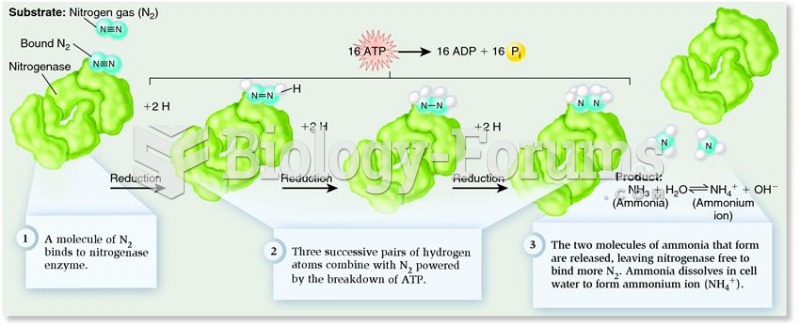
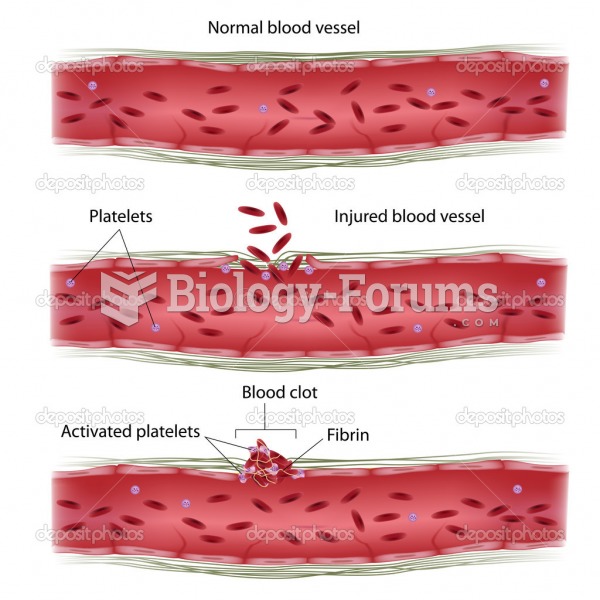
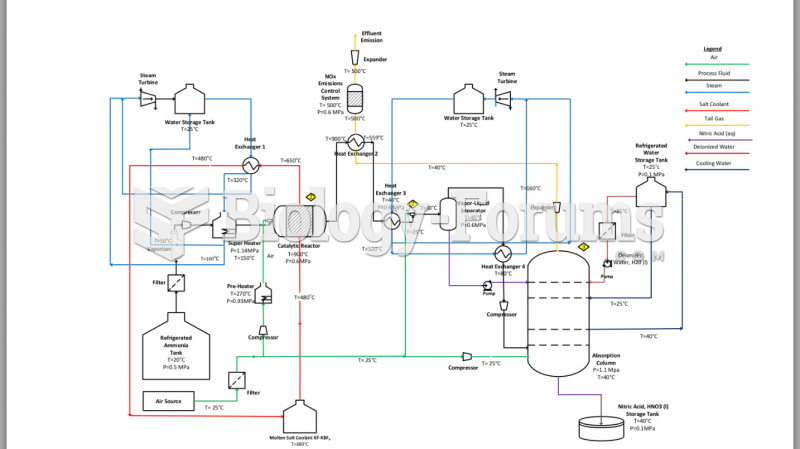
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Disorders that may affect pharmacodynamics include genetic mutations, malnutrition, thyrotoxicosis, myasthenia gravis, Parkinson's disease, and certain forms of insulin-resistant diabetes mellitus.
Did you know?
The average office desk has 400 times more bacteria on it than a toilet.
Did you know?
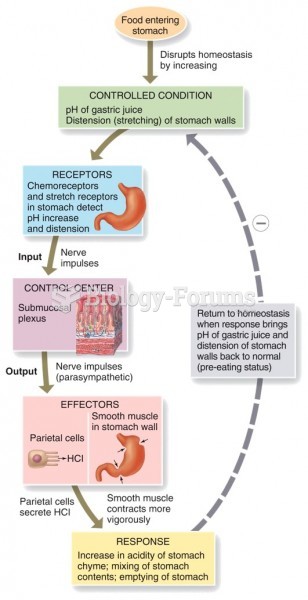
Human stomach acid is strong enough to dissolve small pieces of metal such as razor blades or staples.
Did you know?
Critical care patients are twice as likely to receive the wrong medication. Of these errors, 20% are life-threatening, and 42% require additional life-sustaining treatments.
Did you know?
The B-complex vitamins and vitamin C are not stored in the body and must be replaced each day.