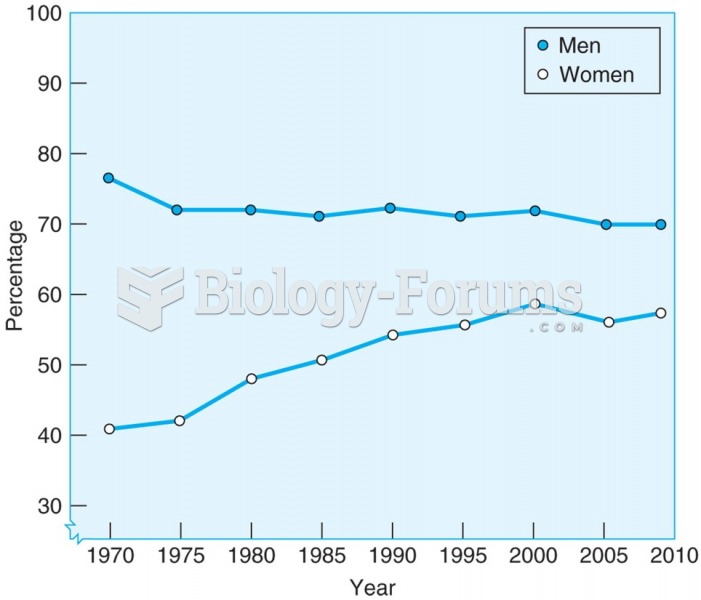
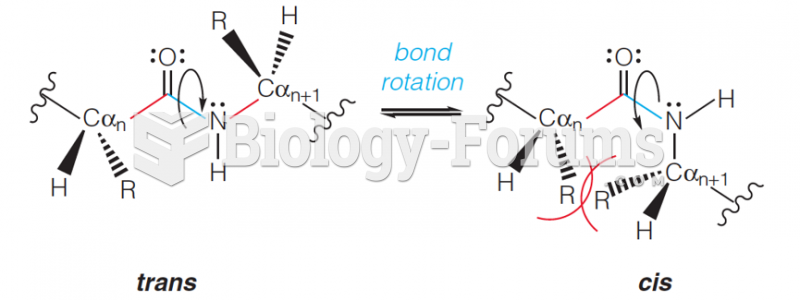
|
|
|
There are 20 feet of blood vessels in each square inch of human skin.
The immune system needs 9.5 hours of sleep in total darkness to recharge completely.
About 60% of newborn infants in the United States are jaundiced; that is, they look yellow. Kernicterus is a form of brain damage caused by excessive jaundice. When babies begin to be affected by excessive jaundice and begin to have brain damage, they become excessively lethargic.
In the United States, there is a birth every 8 seconds, according to the U.S. Census Bureau's Population Clock.
Immunoglobulin injections may give short-term protection against, or reduce severity of certain diseases. They help people who have an inherited problem making their own antibodies, or those who are having certain types of cancer treatments.