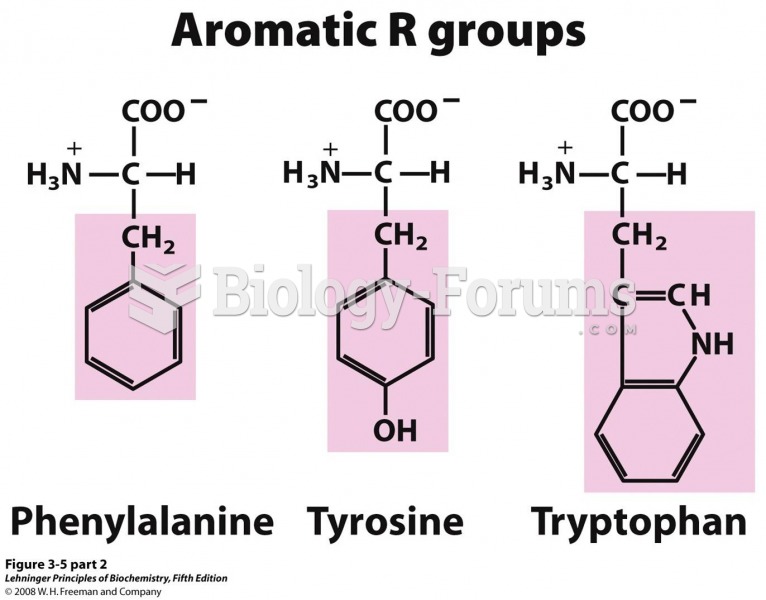
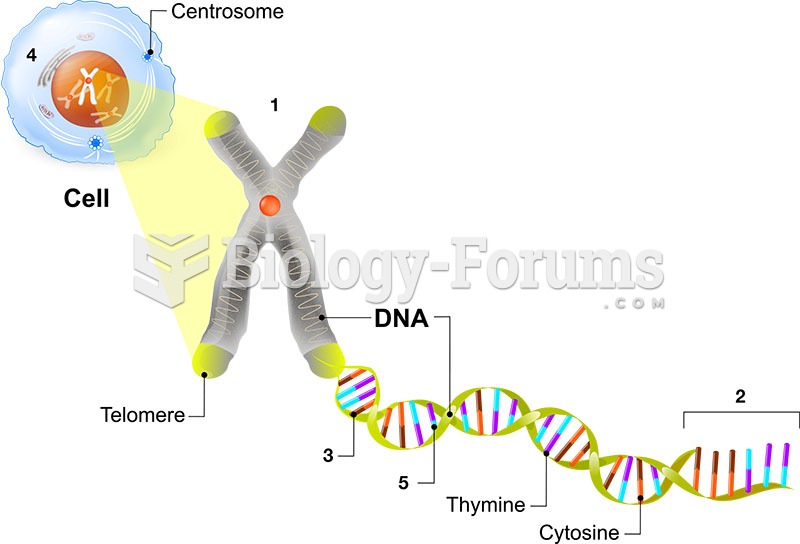
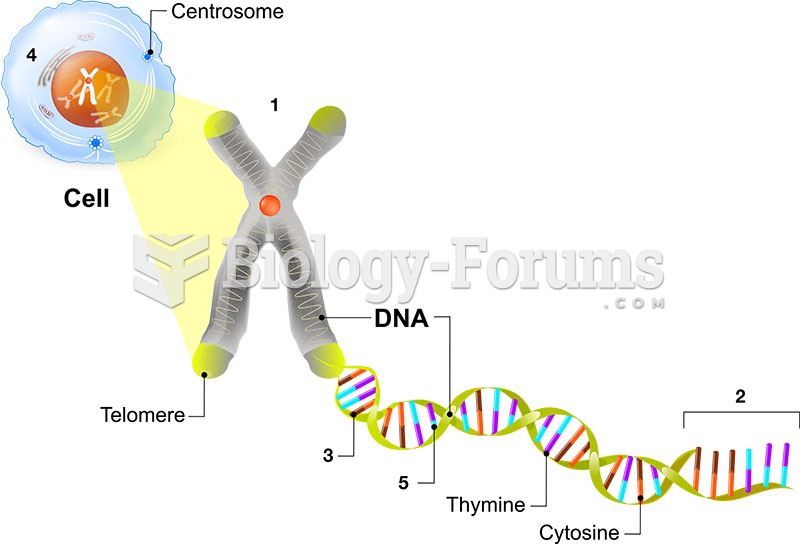
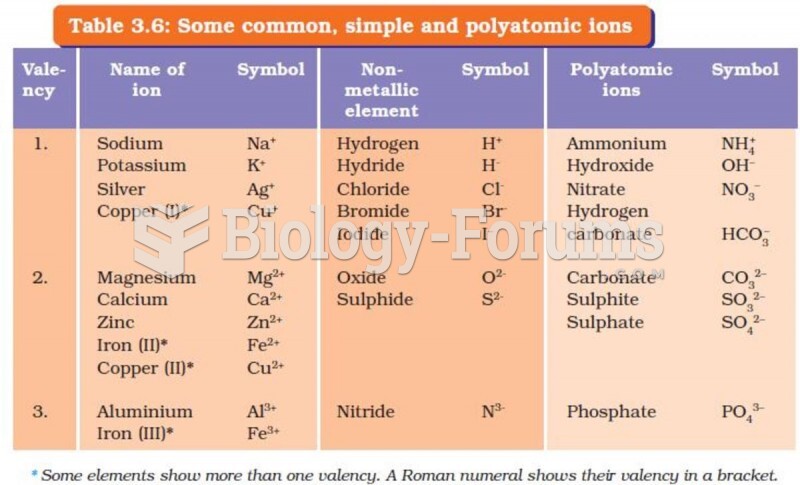
|
|
|
Once thought to have neurofibromatosis, Joseph Merrick (also known as "the elephant man") is now, in retrospect, thought by clinical experts to have had Proteus syndrome. This endocrine disease causes continued and abnormal growth of the bones, muscles, skin, and so on and can become completely debilitating with severe deformities occurring anywhere on the body.
Urine turns bright yellow if larger than normal amounts of certain substances are consumed; one of these substances is asparagus.
Anti-aging claims should not ever be believed. There is no supplement, medication, or any other substance that has been proven to slow or stop the aging process.
A seasonal flu vaccine is the best way to reduce the chances you will get seasonal influenza and spread it to others.
Increased intake of vitamin D has been shown to reduce fractures up to 25% in older people.