|
|
|
The lipid bilayer is made of phospholipids. They are arranged in a double layer because one of their ends is attracted to water while the other is repelled by water.
Illicit drug use costs the United States approximately $181 billion every year.
Coca-Cola originally used coca leaves and caffeine from the African kola nut. It was advertised as a therapeutic agent and "pickerupper." Eventually, its formulation was changed, and the coca leaves were removed because of the effects of regulation on cocaine-related products.
Cutaneous mucormycosis is a rare fungal infection that has been fatal in at least 29% of cases, and in as many as 83% of cases, depending on the patient's health prior to infection. It has occurred often after natural disasters such as tornados, and early treatment is essential.
The largest baby ever born weighed more than 23 pounds but died just 11 hours after his birth in 1879. The largest surviving baby was born in October 2009 in Sumatra, Indonesia, and weighed an astounding 19.2 pounds at birth.
 When using forearm pressure on the top of the shoulder, push with your back foot to increase your ...
When using forearm pressure on the top of the shoulder, push with your back foot to increase your ...
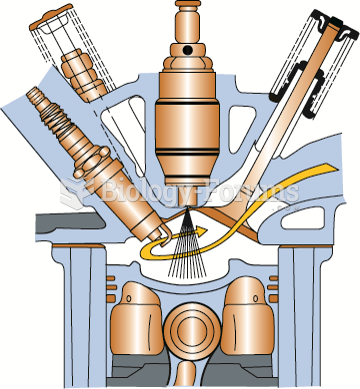 A gasoline direct-injection system injects fuel under high pressure directly into the combustion ...
A gasoline direct-injection system injects fuel under high pressure directly into the combustion ...