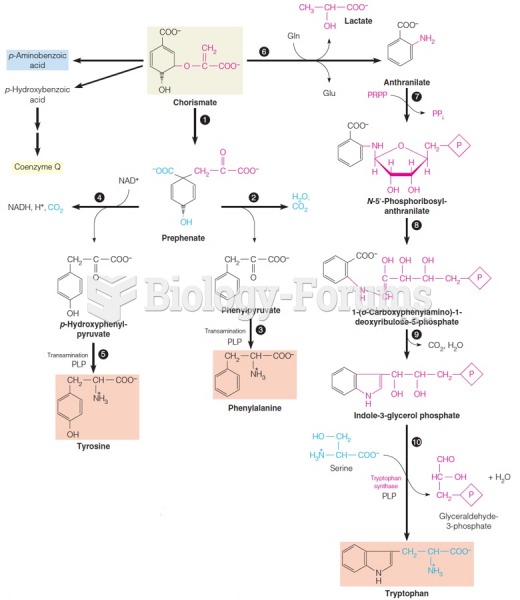
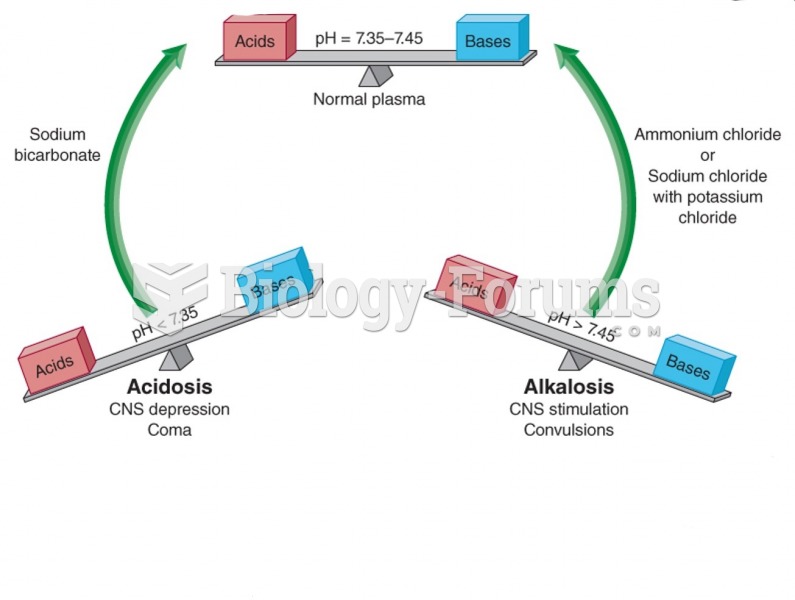
|
|
|
Famous people who died from poisoning or drug overdose include, Adolf Hitler, Socrates, Juan Ponce de Leon, Marilyn Monroe, Judy Garland, and John Belushi.
Hip fractures are the most serious consequences of osteoporosis. The incidence of hip fractures increases with each decade among patients in their 60s to patients in their 90s for both women and men of all populations. Men and women older than 80 years of age show the highest incidence of hip fractures.
There used to be a metric calendar, as well as metric clocks. The metric calendar, or "French Republican Calendar" divided the year into 12 months, but each month was divided into three 10-day weeks. Each day had 10 decimal hours. Each hour had 100 decimal minutes. Due to lack of popularity, the metric clocks and calendars were ended in 1795, three years after they had been first marketed.
On average, someone in the United States has a stroke about every 40 seconds. This is about 795,000 people per year.
In ancient Rome, many of the richer people in the population had lead-induced gout. The reason for this is unclear. Lead poisoning has also been linked to madness.
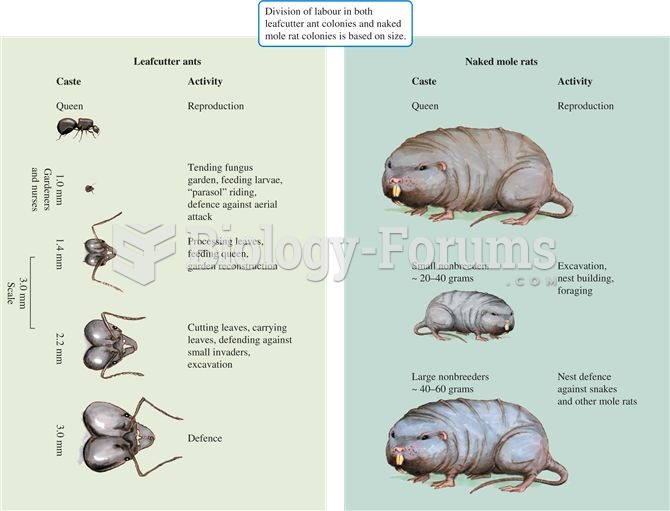 Division of labour among castes of leafcutter ants, Atta sexdens, and naked mole rats, Heterocephalu
Division of labour among castes of leafcutter ants, Atta sexdens, and naked mole rats, Heterocephalu
 At breakfast, a middle-class husband sits absorbed in the newspaper and the public affairs of the da
At breakfast, a middle-class husband sits absorbed in the newspaper and the public affairs of the da