|
|
|
The immune system needs 9.5 hours of sleep in total darkness to recharge completely.
Tobacco depletes the body of vitamins A, C, and E, which can result in any of the following: dry hair, dry skin, dry eyes, poor growth, night blindness, abscesses, insomnia, fatigue, reproductive system problems, sinusitis, pneumonia, frequent respiratory problems, skin disorders, weight loss, rickets, osteomalacia, nervousness, muscle spasms, leg cramps, extremity numbness, bone malformations, decayed teeth, difficulty in walking, irritability, restlessness, profuse sweating, increased uric acid (gout), joint damage, damaged red blood cells, destruction of nerves, infertility, miscarriage, and many types of cancer.
Signs and symptoms that may signify an eye tumor include general blurred vision, bulging eye(s), double vision, a sensation of a foreign body in the eye(s), iris defects, limited ability to move the eyelid(s), limited ability to move the eye(s), pain or discomfort in or around the eyes or eyelids, red or pink eyes, white or cloud spots on the eye(s), colored spots on the eyelid(s), swelling around the eyes, swollen eyelid(s), and general vision loss.
On average, the stomach produces 2 L of hydrochloric acid per day.
After a vasectomy, it takes about 12 ejaculations to clear out sperm that were already beyond the blocked area.
 The paleoanthropologist must understand these deformations in order to figure out which strata a fos
The paleoanthropologist must understand these deformations in order to figure out which strata a fos
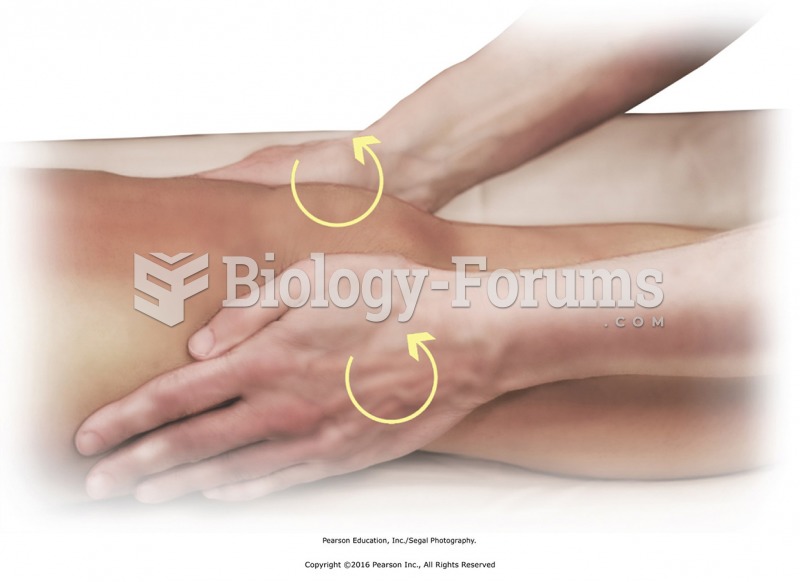 Apply circular friction with the heels of the hands to tendons that cross the knee. Place heels of ...
Apply circular friction with the heels of the hands to tendons that cross the knee. Place heels of ...