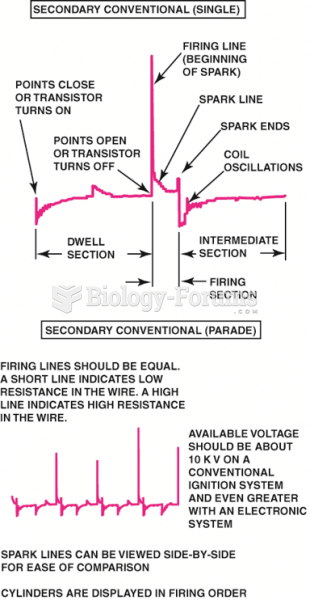
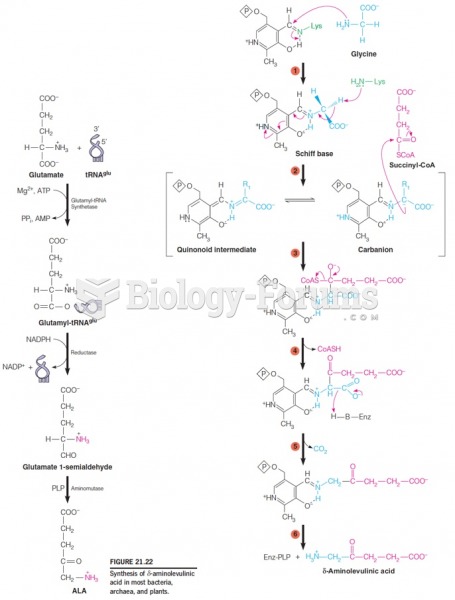
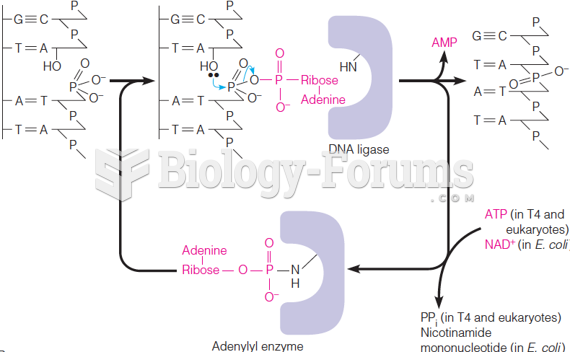
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
The most common treatment options for addiction include psychotherapy, support groups, and individual counseling.
Did you know?
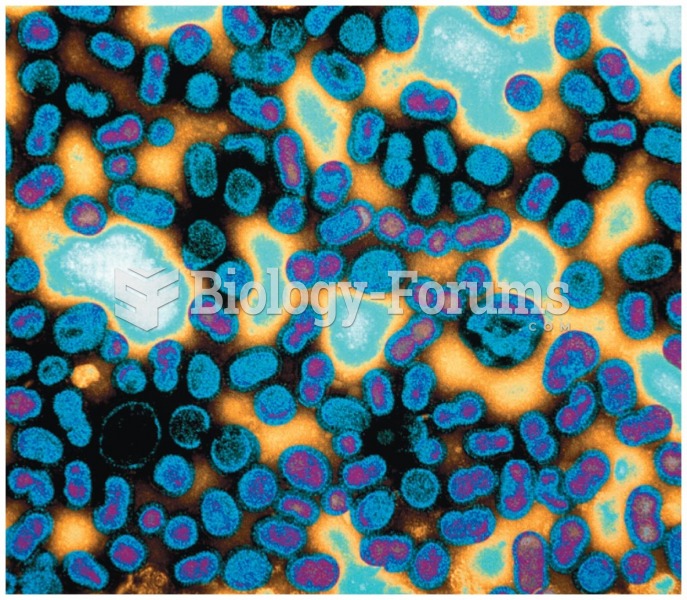
Bacteria have flourished on the earth for over three billion years. They were the first life forms on the planet.
Did you know?
In most cases, kidneys can recover from almost complete loss of function, such as in acute kidney (renal) failure.
Did you know?
The shortest mature adult human of whom there is independent evidence was Gul Mohammed in India. In 1990, he was measured in New Delhi and stood 22.5 inches tall.
Did you know?
To prove that stomach ulcers were caused by bacteria and not by stress, a researcher consumed an entire laboratory beaker full of bacterial culture. After this, he did indeed develop stomach ulcers, and won the Nobel Prize for his discovery.