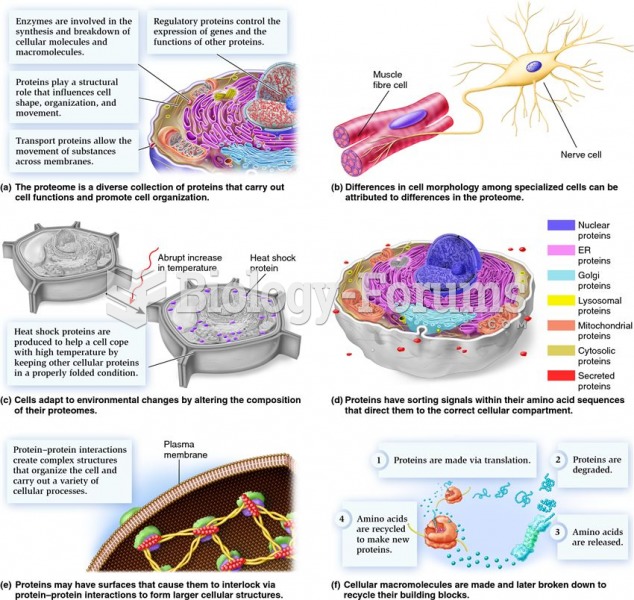
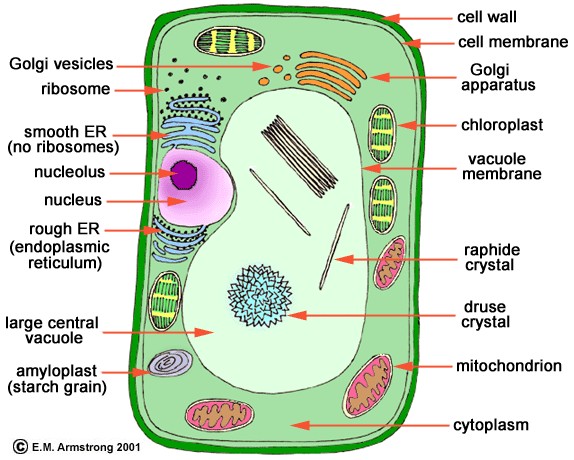
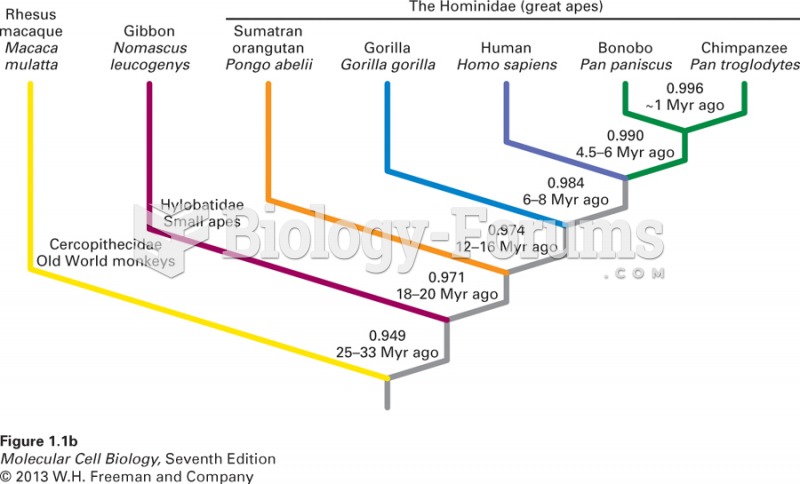
This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
The lipid bilayer is made of phospholipids. They are arranged in a double layer because one of their ends is attracted to water while the other is repelled by water.
Did you know?
Not getting enough sleep can greatly weaken the immune system. Lack of sleep makes you more likely to catch a cold, or more difficult to fight off an infection.
Did you know?
According to the Migraine Research Foundation, migraines are the third most prevalent illness in the world. Women are most affected (18%), followed by children of both sexes (10%), and men (6%).
Did you know?
The average office desk has 400 times more bacteria on it than a toilet.
Did you know?
In 1844, Charles Goodyear obtained the first patent for a rubber condom.