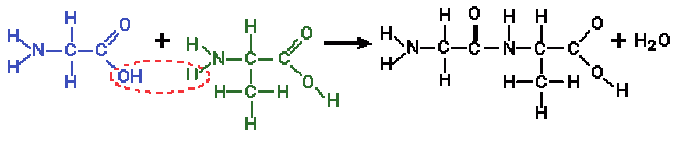
|
|
|
When Gabriel Fahrenheit invented the first mercury thermometer, he called "zero degrees" the lowest temperature he was able to attain with a mixture of ice and salt. For the upper point of his scale, he used 96°, which he measured as normal human body temperature (we know it to be 98.6° today because of more accurate thermometers).
The first oncogene was discovered in 1970 and was termed SRC (pronounced "SARK").
If you use artificial sweeteners, such as cyclamates, your eyes may be more sensitive to light. Other factors that will make your eyes more sensitive to light include use of antibiotics, oral contraceptives, hypertension medications, diuretics, and antidiabetic medications.
The first documented use of surgical anesthesia in the United States was in Connecticut in 1844.
The calories found in one piece of cherry cheesecake could light a 60-watt light bulb for 1.5 hours.