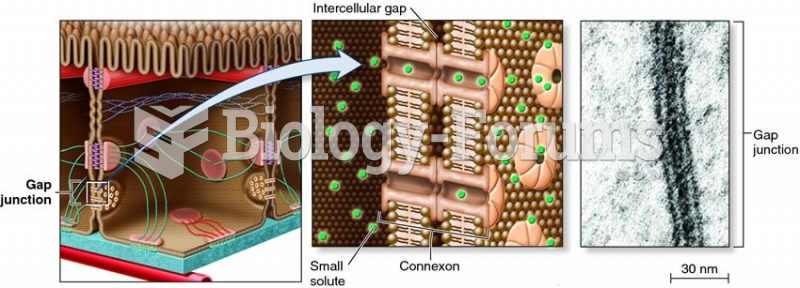
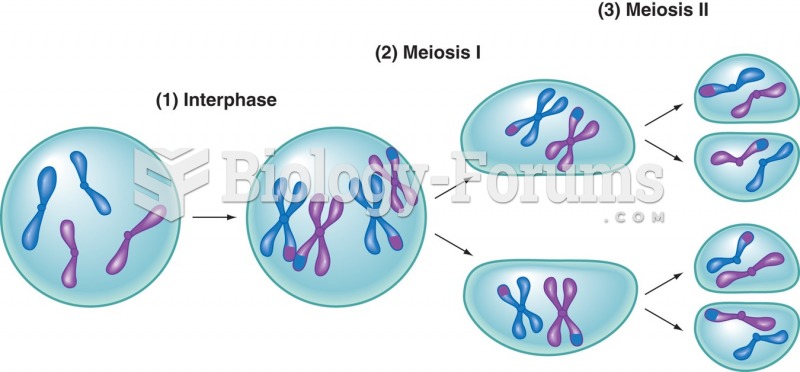
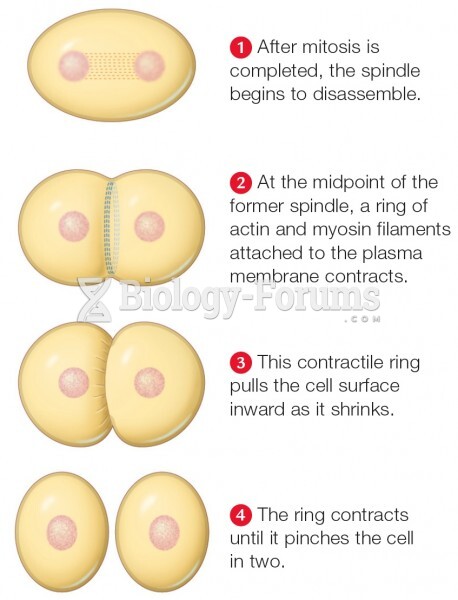
|
|
|
Your chance of developing a kidney stone is 1 in 10. In recent years, approximately 3.7 million people in the United States were diagnosed with a kidney disease.
People about to have surgery must tell their health care providers about all supplements they take.
An identified risk factor for osteoporosis is the intake of excessive amounts of vitamin A. Dietary intake of approximately double the recommended daily amount of vitamin A, by women, has been shown to reduce bone mineral density and increase the chances for hip fractures compared with women who consumed the recommended daily amount (or less) of vitamin A.
Cancer has been around as long as humankind, but only in the second half of the twentieth century did the number of cancer cases explode.
If all the neurons in the human body were lined up, they would stretch more than 600 miles.