|
|
|
Adults are resistant to the bacterium that causes Botulism. These bacteria thrive in honey – therefore, honey should never be given to infants since their immune systems are not yet resistant.
The first-known contraceptive was crocodile dung, used in Egypt in 2000 BC. Condoms were also reportedly used, made of animal bladders or intestines.
Sperm cells are so tiny that 400 to 500 million (400,000,000–500,000,000) of them fit onto 1 tsp.
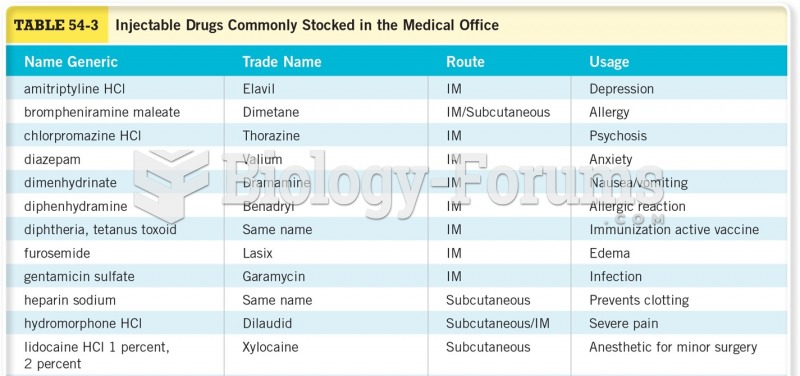
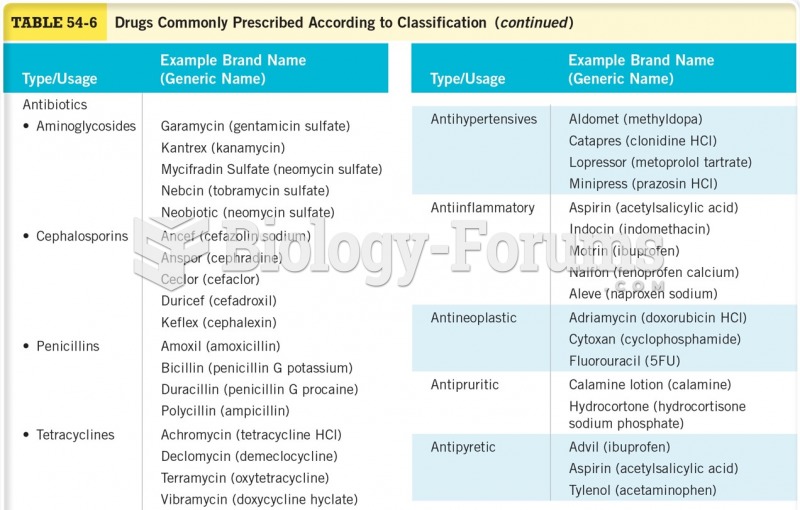
The use of salicylates dates back 2,500 years to Hippocrates's recommendation of willow bark (from which a salicylate is derived) as an aid to the pains of childbirth. However, overdosage of salicylates can harm body fluids, electrolytes, the CNS, the GI tract, the ears, the lungs, the blood, the liver, and the kidneys and cause coma or death.
Multiple experimental evidences have confirmed that at the molecular level, cancer is caused by lesions in cellular DNA.
 Carnivorous plants, such as this pitcher plant (Sarracenia purpurea) are commonly found in bog habit
Carnivorous plants, such as this pitcher plant (Sarracenia purpurea) are commonly found in bog habit
 Killer whales, like this one spotted near Alaska, commonly breach, often lifting their entire bodies
Killer whales, like this one spotted near Alaska, commonly breach, often lifting their entire bodies