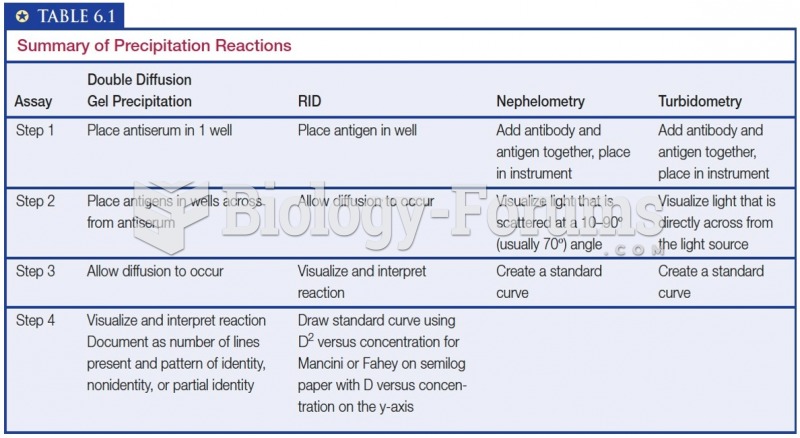
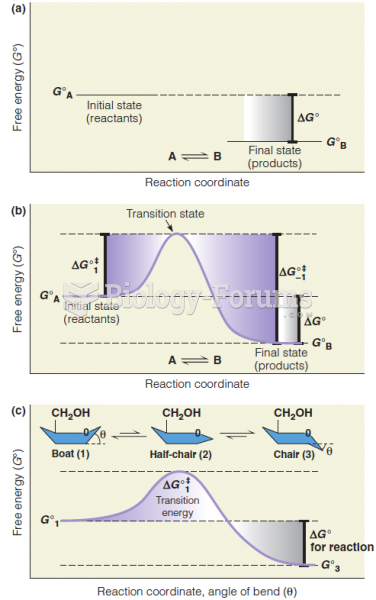
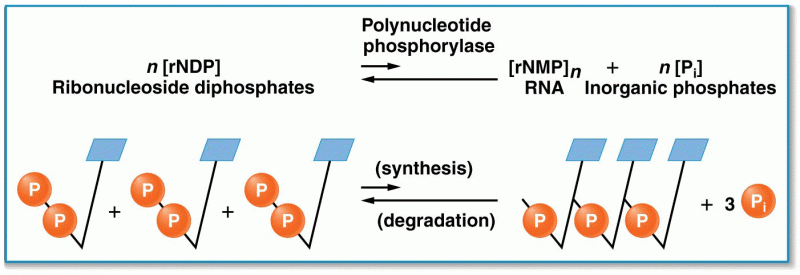
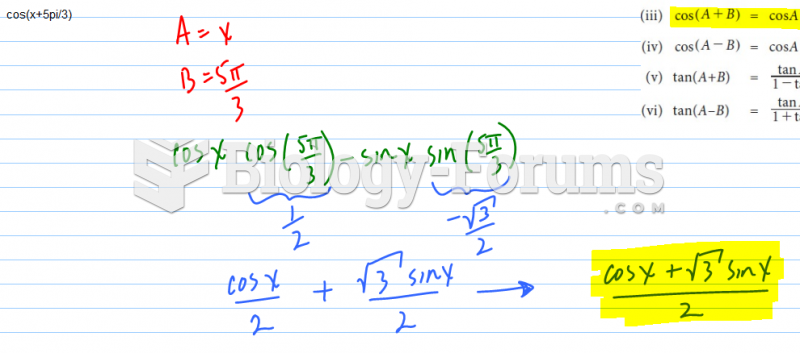This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
Approximately 500,000 babies are born each year in the United States to teenage mothers.
Did you know?
Normal urine is sterile. It contains fluids, salts, and waste products. It is free of bacteria, viruses, and fungi.
Did you know?
The top five reasons that children stay home from school are as follows: colds, stomach flu (gastroenteritis), ear infection (otitis media), pink eye (conjunctivitis), and sore throat.
Did you know?
The immune system needs 9.5 hours of sleep in total darkness to recharge completely.
Did you know?
The first-known contraceptive was crocodile dung, used in Egypt in 2000 BC. Condoms were also reportedly used, made of animal bladders or intestines.