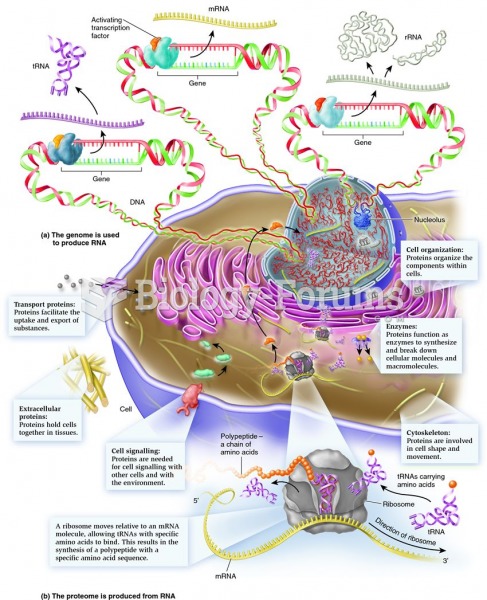
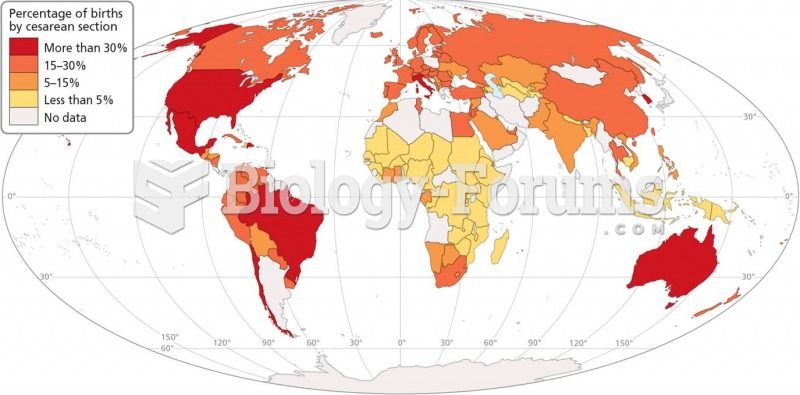
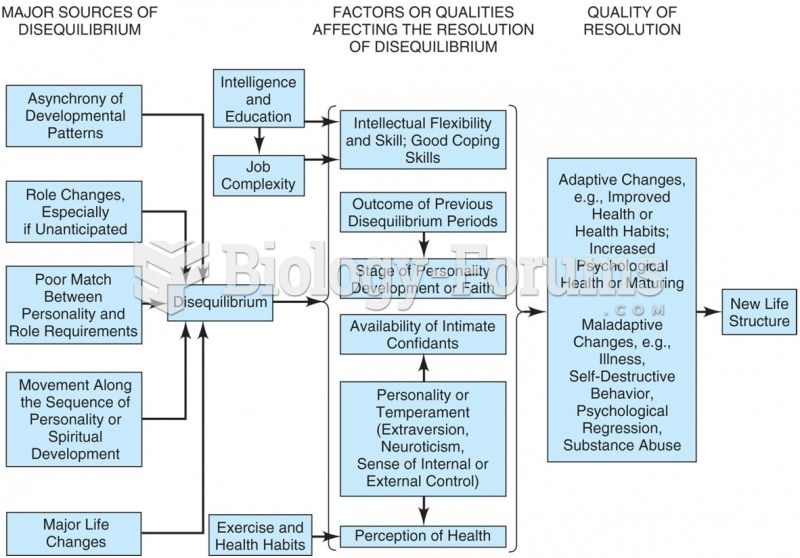
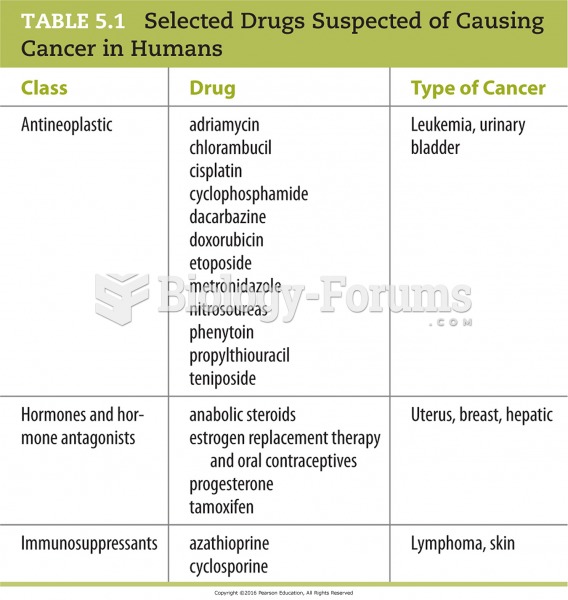
|
|
|
According to the CDC, approximately 31.7% of the U.S. population has high low-density lipoprotein (LDL) or "bad cholesterol" levels.
For pediatric patients, intravenous fluids are the most commonly cited products involved in medication errors that are reported to the USP.
More than 2,500 barbiturates have been synthesized. At the height of their popularity, about 50 were marketed for human use.
Many medications that are used to treat infertility are injected subcutaneously. This is easy to do using the anterior abdomen as the site of injection but avoiding the area directly around the belly button.
Drugs are in development that may cure asthma and hay fever once and for all. They target leukotrienes, which are known to cause tightening of the air passages in the lungs and increase mucus productions in nasal passages.