This topic contains a solution. Click here to go to the answer
|
|
|
Did you know?
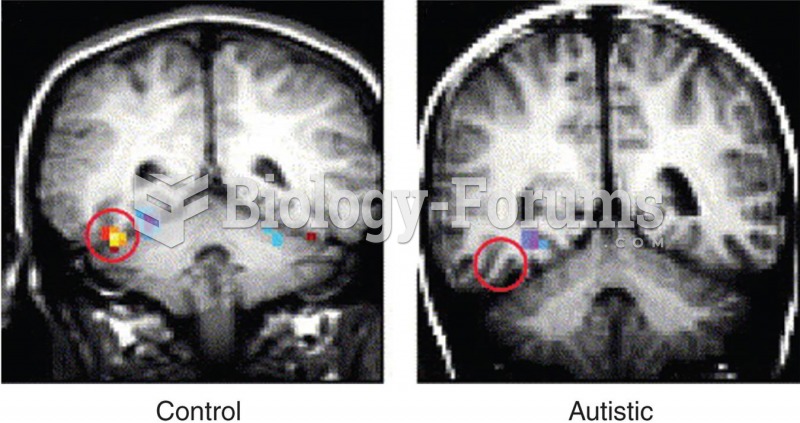
The first oncogene was discovered in 1970 and was termed SRC (pronounced "SARK").
Did you know?
The Romans did not use numerals to indicate fractions but instead used words to indicate parts of a whole.
Did you know?
Your heart beats over 36 million times a year.
Did you know?
About 600,000 particles of skin are shed every hour by each human. If you live to age 70 years, you have shed 105 pounds of dead skin.
Did you know?
Your chance of developing a kidney stone is 1 in 10. In recent years, approximately 3.7 million people in the United States were diagnosed with a kidney disease.