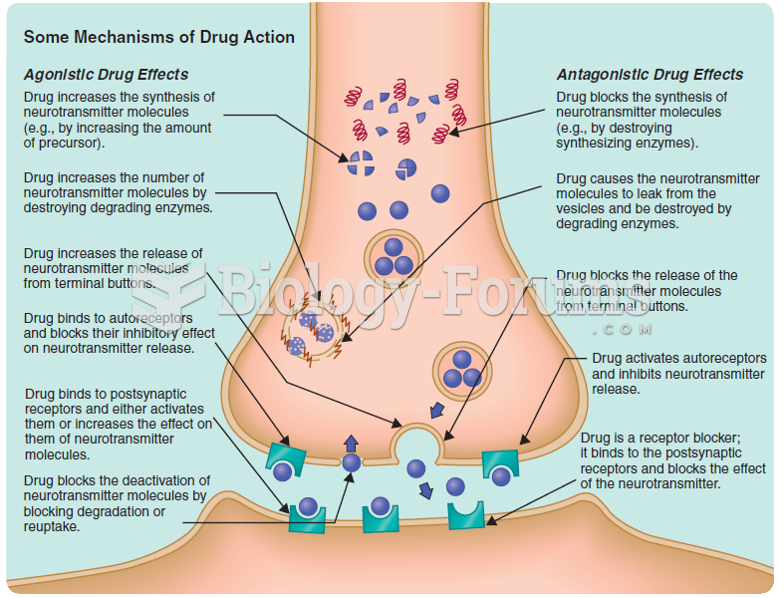
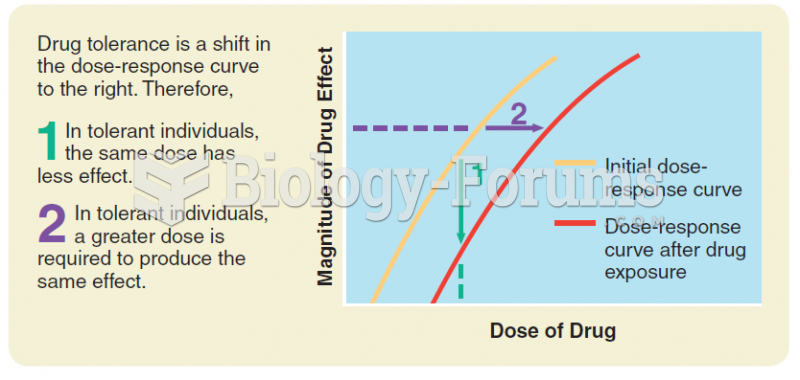
Answer to Question 1
Correct Answer: 4
Rationale 1: Post-marketing studies take place after clinical trials to check for any new harmful effects in a larger and more diverse population.
Rationale 2: Clinical investigation is the second and longest part of the drug approval process, and takes place in three different phases, termed clinical phase trials. During this phase, clinical investigators address concerns such as whether the drug worsens other medical conditions; establish drug doses; and try to identify adverse effects.
Rationale 3: Postclinical investigation is not a phase of the approval process.
Rationale 4: Preclinical investigation involves basic science research.
Global Rationale: Preclinical investigation involves basic science research. Post-marketing studies take place after clinical trials to check for any new harmful effects in a larger and more diverse population. Clinical investigation is the second and longest part of the drug approval process, and takes place in three different phases, termed clinical phase trials. During this phase, clinical investigators address concerns such as whether the drug worsens other medical conditions; establish drug doses; and try to identify adverse effects. Postclinical investigation is not a phase of the approval process.
Answer to Question 2
Correct Answer: 4
Rationale 1: A review of the NDA is the third stage of drug approval. During this stage, clinical phase III trials and animal testing may continue, depending on the results obtained from preclinical testing. If the NDA is approved, the process continues to the final stage. If the NDA is rejected, the process stops until concerns are addressed.
Rationale 2: During these phases, clinical pharmacologists, researchers, and health care providers examine data from volunteers and large groups of selected patients with certain diseases.
Rationale 3: Post-marketing surveillance is the fourth stage of the drug approval process. It takes place after clinical trials and the NDA review process have been completed. Testing in humans is continued to check for any new harmful effects in larger and more diverse populations.
Rationale 4: Preclinical investigation involves basic science research.
Global Rationale: Preclinical investigation involves basic science research. A review of the NDA is the third stage of drug approval. During this stage, clinical phase III trials and animal testing may continue, depending on the results obtained from preclinical testing. If the NDA is approved, the process continues to the final stage. If the NDA is rejected, the process stops until concerns are addressed. During these phases, clinical pharmacologists, researchers, and health care providers examine data from volunteers and large groups of selected patients with certain diseases.