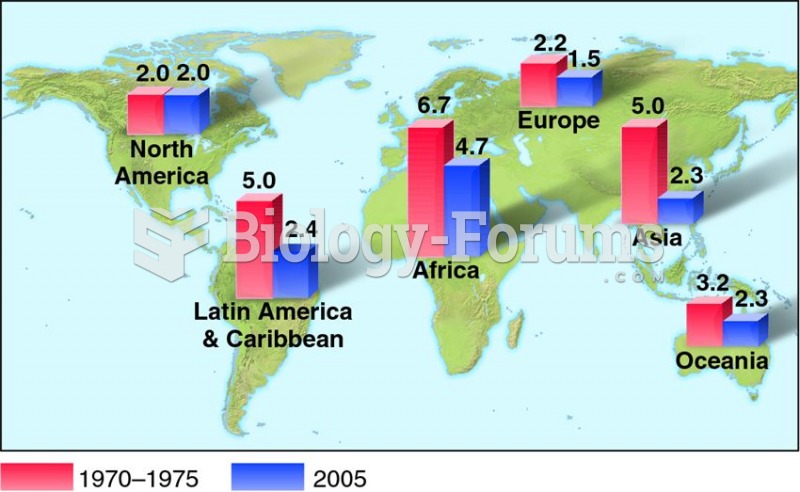
|
|
|
Cutaneous mucormycosis is a rare fungal infection that has been fatal in at least 29% of cases, and in as many as 83% of cases, depending on the patient's health prior to infection. It has occurred often after natural disasters such as tornados, and early treatment is essential.
It is difficult to obtain enough calcium without consuming milk or other dairy foods.
Earwax has antimicrobial properties that reduce the viability of bacteria and fungus in the human ear.
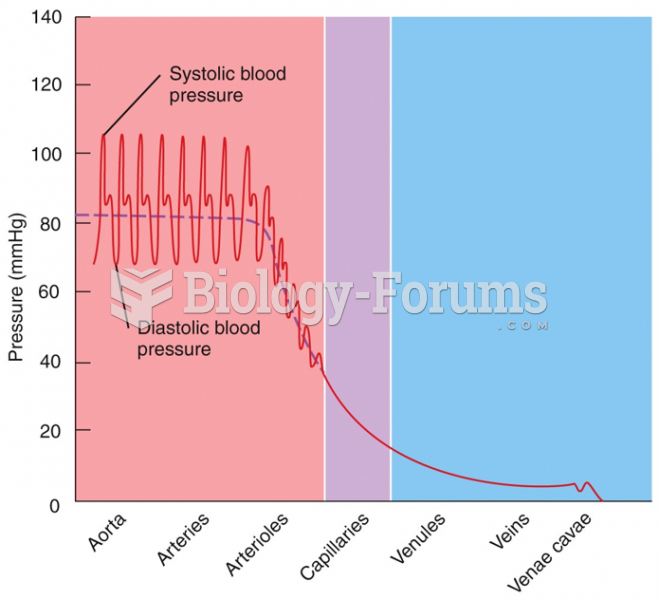
The familiar sounds of your heart are made by the heart's valves as they open and close.
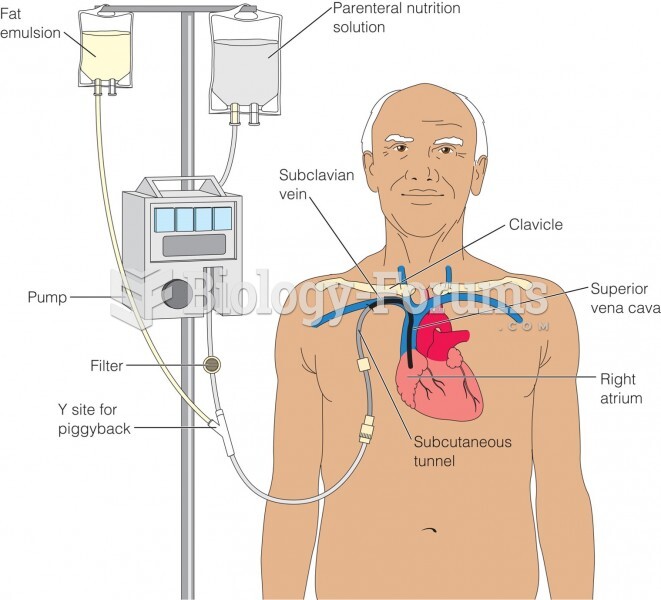
The average person is easily confused by the terms pharmaceutics and pharmacology, thinking they are one and the same. Whereas pharmaceutics is the science of preparing and dispensing drugs (otherwise known as the science of pharmacy), pharmacology is the study of medications.