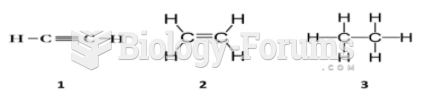Are the following structures chiral as drawn? When placed in solution at 298 K, which structures will show
an optical rotation? Explain.
 Question 2
Question 2DMxt+7nRbmnv8be/avwYs1wlGvTDaFfPY/JXpqE/VToicXWWXNxKKQaaht8F9JxbdMEPFK5GT7aF4/4Q/PQpXIkyrX7CPtuqrYJRmT1+ih0eiE+3HhN/aivziIQuxWz5h5CSLXaZvyprmPLO89j2Kuj8WTTerij6pP48piadgEER6J6/UI4vfBD6KZwtgEy3ovdOgtzD4WgWu0StxcAiYcwv09tVOi4CGdjN2NUoxKo/OQinGQTaOyzB7hL37bEy95Ak9KVcF/HYXhlUG+8/stlM6VqfmYGFCs/mcM6DVf/GI+mdzbCuK0xOPrpo6ja4i1su35L8tFjxQqe6pYprsk4sXgUXvs5Y4699vNL6NRjOv65NQ8HR1ZH/UKnsfDDjfDGLOyfQk/iUaz/IwqIKIvCBgXDdfw5ZTo2nj2Ny6WbovugEeheHriw9m18sD1WtQ7yFCmLCFzHlh/3a98eAyrOfUg8uh4ZzVD49mRryCoUdZ6fg+VrN+Dbp4sBp5diyNBvcT5NPZAHRcpGANe34Mf92rdHoeL4NRFH1/+BKESgrGEw3MSuyR3xzKJjqDPmVTzdviUaFgbOrFyEXXIoaOwdx9lNb5jjZfE7MLn3RPwdG4nuX/+F335agtH1w8wSoPmZWVjM/2gO62u/YmS3N/Fn4v3S43OsAAAG4ElEQVQYOqg7evZrDvz+Gp758D9kKKf1WDEPptGvWXDNiwpPvYfJjxaSD0W2egfffT0GDfLfeidPEWSw/h/hDdbvp0LPeRy8BCBvGPIZalAAdw8ejZ5d+6NLzVDEnTmKC4kE+TrOx9w2cgWHhCMEQNTBC7c6tVHj6Y8OIZB4/iAymiGfmR1xeRFeIDeAQqjbsga4h+vmX8uw64YqIhgh4bIlcPCCie1LPaKvdiCQiPMZgwFhajDE7cTszw8AKIpqdxZAUPi9mLDsayz6cS7aFmGWGns7gPXMI4lZednN/cvx3QkWXxHN6kUACEFkMTVjZCZL87PMeFj9Zgbr2H++wo9UouWKwebFc7HglzPgtLF/zWZckgs0PVasYsqbZnC1+k5wCDJY/0F4g/VzVvLDFIzccie0oMfsrZQbpR4bgdHX3sWUp5vh24b3IcFgTbn9FHDrnVzqPX11GoHg3FKYgTDGN2tueSOKIBRAXEJMJnu5ek83RVbMHPklOGMwKDiBhHM4GMUcUpCYwrbJi1IPPYmnjDO91WYae2NQvPE5Ky9Lvnwc0SQlVygK5jM98sGURs3PTBGx/D0r1inXzuE6X0gFIstXQrUaL2Lpry8iqEBlFFGDQ48Vy5DKO1lxtfHCLV6lALb1tGvvG/Qkrs3WzbmFlkXdOyhhxiJRmUvSL+PHQQ3RsPfb2NN8Nr5+uxtqmNnGnpYYKzU8JWuXkRofN1Oao7MPLVsXGc2QCNUM5iqclnA9Q6tWvDpKUbkjUxoSY6nhKYnaZQw/qpv6ajcCoSibMRgQqwZD3qIoQwUBrmD7ptNmNJoae7vhdfeDZnhZ7gJFIfU6IgE3VJtaoEPzMwvAmPvZDNZ5IstADpWb0chVrQmaN28u/5reUx5hcnbUY8UclJl+M4NrpvumX9ISkcH6a8MbrN8/hZ6QO9GiVVkg5gyuptxCNHYXFiw/Kb+ERYYhLeogjkgRHrdXwFz7Xj2NGBTGQ62rw7yV3LSF9HdLCITc2QIZzXAVqhluP5uGJKlluIkT24/L+1V6P4OGBkE0BVdPxwCFH0Lr6rolbuPm6KcQ3NmiFcoiBmfUYIi4B889WUZmdOC9V/HVUQqXSTi5bhH+uEjxVGPvKMpue94MLwuv3REtpBnyOH7bcx1Iu4YjhzJcPkVKUqaxpvmZAy1jBuuw+t3xWFHmsQfvv/09TielIfbfRXj9rd9xVfoy67FiE+H/t3MGIW1DYRx/OwykvShswpBduw48iLTOsUOrt82x0rHtVNlph0EvBaVCRfEwkDE8zW3s6M4i7lCHK8jmQKvQuWktRYbGg3Vl66RV0pKY/0ibVrtWm0hnW/qdmqTvvbz3++f754W8fEW4ynUk5QWAJBwtL0m3JfxmGeu/zSpi/Sd+d1/lfySDo2jXGzGymsz0VODwzn5ZxgzG9LjW48RT08X0foO5H76onA9GxOa4CTrDAJYPqnyANdG9JIKj7dAbR5CVAX+8eHTVANuTh+i6P4g3r/phadLD6HiLtf1jgxI3MW7SwTCwDJLiGJezbCaDGG3XwziyCiUacLi3hBd3W3LxcMVwC49ffkU8HQbE/iyY/1edAi+DiMgHN27oGZjOgJtdPXjQdQmMNaD5+h143m8rucnIz7RqUoz1r8/P0NOSXimRZtzSPYiPu2KmafIpVYjzuYqIzj/HvWaFaVM3XK/n8TOHdBwmnQEDFboJM1UjqspCCSx6OtExFDi6aR4msLXix3cuDpnv4f4WAv417PBKArxUCGNWM1xzMUpOWC5NE4vwdHZgKKBMXcQ9bEcO0nyFWBj++QWs7fAFvFOhMVjNLszFKDlhOaRILHrQ2TGErAyZNgXEwn58+vIN23HFcQAQ+3IQL2cbRbxM9i8+guBSAD/2BPC7HKLZGW321ORnWRIafouzhhjH1soC/KFo7sFBbpRiRS3aE7gWVE8hNGaF2TWHSll/DU965CuSw2SfA+4prnRWZiGC2eFeOCc28i7qAk3ogGYCKW4SfQ43pjh1ubGFyCyGe52Y2PjXxTWfmirkCKTATfbB4Z7CaTIQ+xyw6trQ4mVyz8nPzq6fStYUKxoRl+QqIDI7jF7nBCpp/RfkYZV8Z1fNBaQEW/f6mGS1s9bcepHCDifD02yGtzBbW2ORz6sLy9MRbQSkxDrz+iRmtbeyU2SQV5+z8PQM4y021tZYm0vKtJE5z9ISS6x7mU+yMnvRYCD256mG5nOp9DK5XfIzzXTzK5RkTbGSD0zl3mlck2E2PcMzi62NVdL6a3/So1ILKkYEiAARIAJEgAjUNwF61K5v/Wn0RIAIEAEiQATqhgBNeupGahooESACRIAIEIH6JvAXBRIdpmCJi7sAAAAASUVORK5CYII= />
Analyzing the planar structures reveals that compounds (b)
Question 3
Will the following compound show any optical activity if there is restricted rotation along the central C-C
bond? What will happen to the optical activity at elevated temperatures as the rotation becomes less restricted.
 Question 4
Question 4Which of the following compounds are chiral? Which, if any, are meso? Which, if any, does not have apossible diastereomer?

Question 5A long polymer chain, such as polyethylene (-CH2CH2-)n, can potentially exist in solution as a chiral object.
Give two examples of chiral structures that a polyethylene chain could adopt.
Question 6One reason we can be sure that sp3-hybridized carbon atoms are tetrahedral is the number of stereoisomers
that can exist for different organic compounds.
(a) How many stereoisomers are possible for CHCl3, CH2Cl2, and CHClBrF if the four bonds to carbon have a
tetrahedral arrangement?
(b) How many stereoisomers would be possible for each of these compounds if the four bonds to the carbon had a square
planar geometry?
Question 7Think about the helical coil of a telephone cord or a spiral binding and suppose that you view the spiral from
one end and find that it is a left-handed twist. If you view the same spiral from the other end, is it a right-handed or a lefthanded twist?