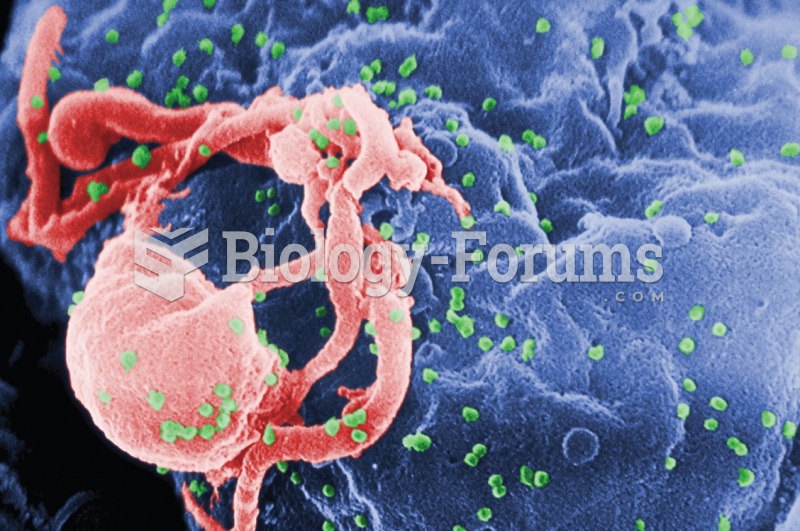
|
|
|
Opium has influenced much of the world's most popular literature. The following authors were all opium users, of varying degrees: Lewis Carroll, Charles, Dickens, Arthur Conan Doyle, and Oscar Wilde.
Most women experience menopause in their 50s. However, in 1994, an Italian woman gave birth to a baby boy when she was 61 years old.
Many supplement containers do not even contain what their labels say. There are many documented reports of products containing much less, or more, that what is listed on their labels. They may also contain undisclosed prescription drugs and even contaminants.
Russia has the highest death rate from cardiovascular disease followed by the Ukraine, Romania, Hungary, and Poland.
According to the FDA, adverse drug events harmed or killed approximately 1,200,000 people in the United States in the year 2015.
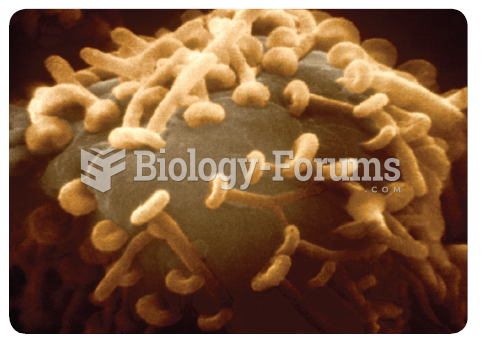 A color-enhanced scanning electron micrograph of a neuron cell body (grey) studded with terminal ...
A color-enhanced scanning electron micrograph of a neuron cell body (grey) studded with terminal ...
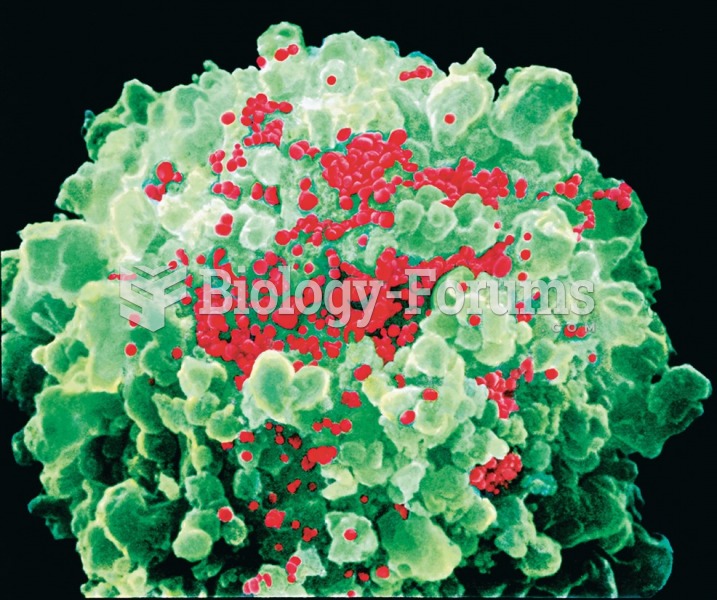 Colored scanning electron micrograph (SEM) of a helper T cell (green) with HIV viruses (red) budding ...
Colored scanning electron micrograph (SEM) of a helper T cell (green) with HIV viruses (red) budding ...